You are browsing environment: FUNGIDB
CAZyme Information: QRC91113.1
You are here: Home > Sequence: QRC91113.1
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Parastagonospora nodorum | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Ascomycota; Dothideomycetes; ; Phaeosphaeriaceae; Parastagonospora; Parastagonospora nodorum | |||||||||||
| CAZyme ID | QRC91113.1 | |||||||||||
| CAZy Family | AA3 | |||||||||||
| CAZyme Description | Levanase [Source:UniProtKB/TrEMBL;Acc:A0A7U2ESB5] | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | ||||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH32 | 27 | 338 | 1e-66 | 0.9658703071672355 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| 350134 | GH32_Inu-like | 7.86e-114 | 34 | 335 | 3 | 288 | glycoside hydrolase family 32 protein such as Aspergillus ficuum endo-inulinase (Inu2). This subfamily of glycosyl hydrolase family GH32 includes endo-inulinase (inu2, EC 3.2.1.7), exo-inulinase (Inu1, EC 3.2.1.80), invertase (EC 3.2.1.26), and levan fructotransferase (LftA, EC 4.2.2.16), among others. These enzymes cleave sucrose into fructose and glucose via beta-fructofuranosidase activity, producing invert sugar that is a mixture of dextrorotatory D-glucose and levorotatory D-fructose, thus named invertase (EC 3.2.1.26). These retaining enzymes (i.e. they retain the configuration at anomeric carbon atom of the substrate) catalyze hydrolysis in two steps involving a covalent glycosyl enzyme intermediate: an aspartate located close to the N-terminus acts as the catalytic nucleophile and a glutamate acts as the general acid/base; a conserved aspartate residue in the Arg-Asp-Pro (RDP) motif stabilizes the transition state. These enzymes are predicted to display a 5-fold beta-propeller fold as found for GH43 and CH68. The breakdown of sucrose is widely used as a carbon or energy source by bacteria, fungi, and plants. Invertase is used commercially in the confectionery industry, since fructose has a sweeter taste than sucrose and a lower tendency to crystallize. A common structural feature of all these enzymes is a 5-bladed beta-propeller domain, similar to GH43, that contains the catalytic acid and catalytic base. A long V-shaped groove, partially enclosed at one end, forms a single extended substrate-binding surface across the face of the propeller. |
| 224536 | SacC | 2.75e-87 | 23 | 516 | 29 | 483 | Sucrose-6-phosphate hydrolase SacC, GH32 family [Carbohydrate transport and metabolism]. |
| 214757 | Glyco_32 | 6.71e-86 | 27 | 480 | 1 | 436 | Glycosyl hydrolases family 32. |
| 395193 | Glyco_hydro_32N | 7.70e-69 | 27 | 349 | 1 | 308 | Glycosyl hydrolases family 32 N-terminal domain. This domain corresponds to the N-terminal domain of glycosyl hydrolase family 32 which forms a five bladed beta propeller structure. |
| 350110 | GH32_FFase | 1.92e-47 | 34 | 334 | 2 | 279 | Glycosyl hydrolase family 32, beta-fructosidases. Glycosyl hydrolase family GH32 cleaves sucrose into fructose and glucose via beta-fructofuranosidase activity, producing invert sugar that is a mixture of dextrorotatory D-glucose and levorotatory D-fructose, thus named invertase (EC 3.2.1.26). This family also contains other fructofuranosidases such as inulinase (EC 3.2.1.7), exo-inulinase (EC 3.2.1.80), levanase (EC 3.2.1.65), and transfructosidases such sucrose:sucrose 1-fructosyltransferase (EC 2.4.1.99), fructan:fructan 1-fructosyltransferase (EC 2.4.1.100), sucrose:fructan 6-fructosyltransferase (EC 2.4.1.10), fructan:fructan 6G-fructosyltransferase (EC 2.4.1.243) and levan fructosyltransferases (EC 2.4.1.-). These retaining enzymes (i.e. they retain the configuration at anomeric carbon atom of the substrate) catalyze hydrolysis in two steps involving a covalent glycosyl enzyme intermediate: an aspartate located close to the N-terminus acts as the catalytic nucleophile and a glutamate acts as the general acid/base; a conserved aspartate residue in the Arg-Asp-Pro (RDP) motif stabilizes the transition state. These enzymes are predicted to display a 5-fold beta-propeller fold as found for GH43 and CH68. The breakdown of sucrose is widely used as a carbon or energy source by bacteria, fungi, and plants. Invertase is used commercially in the confectionery industry, since fructose has a sweeter taste than sucrose and a lower tendency to crystallize. A common structural feature of all these enzymes is a 5-bladed beta-propeller domain, similar to GH43, that contains the catalytic acid and catalytic base. A long V-shaped groove, partially enclosed at one end, forms a single extended substrate-binding surface across the face of the propeller. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| 0.0 | 1 | 520 | 1 | 520 | |
| 7.42e-264 | 5 | 516 | 19 | 528 | |
| 2.74e-263 | 9 | 516 | 22 | 528 | |
| 7.28e-263 | 9 | 516 | 23 | 529 | |
| 4.51e-262 | 9 | 516 | 22 | 528 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 1.17e-72 | 26 | 506 | 4 | 466 | Crystal Structure of Levan Fructotransferase from Arthrobacter ureafaciens [Paenarthrobacter ureafaciens],4FFF_B Crystal Structure of Levan Fructotransferase from Arthrobacter ureafaciens [Paenarthrobacter ureafaciens],4FFF_C Crystal Structure of Levan Fructotransferase from Arthrobacter ureafaciens [Paenarthrobacter ureafaciens],4FFF_D Crystal Structure of Levan Fructotransferase from Arthrobacter ureafaciens [Paenarthrobacter ureafaciens] |
|
| 1.23e-72 | 26 | 506 | 4 | 466 | Crystal Structure of Levan Fructotransferase from Arthrobacter ureafaciens in complex with DFA-IV [Paenarthrobacter ureafaciens],4FFG_B Crystal Structure of Levan Fructotransferase from Arthrobacter ureafaciens in complex with DFA-IV [Paenarthrobacter ureafaciens],4FFG_C Crystal Structure of Levan Fructotransferase from Arthrobacter ureafaciens in complex with DFA-IV [Paenarthrobacter ureafaciens],4FFG_D Crystal Structure of Levan Fructotransferase from Arthrobacter ureafaciens in complex with DFA-IV [Paenarthrobacter ureafaciens] |
|
| 6.64e-72 | 26 | 506 | 4 | 466 | Crystal Structure of Levan Fructotransferase D54N mutant from Arthrobacter ureafaciens in complex with sucrose [Paenarthrobacter ureafaciens],4FFH_B Crystal Structure of Levan Fructotransferase D54N mutant from Arthrobacter ureafaciens in complex with sucrose [Paenarthrobacter ureafaciens],4FFH_C Crystal Structure of Levan Fructotransferase D54N mutant from Arthrobacter ureafaciens in complex with sucrose [Paenarthrobacter ureafaciens],4FFH_D Crystal Structure of Levan Fructotransferase D54N mutant from Arthrobacter ureafaciens in complex with sucrose [Paenarthrobacter ureafaciens],4FFI_A Crystal Structure of Levan Fructotransferase D54N mutant from Arthrobacter ureafaciens in complex with levanbiose [Paenarthrobacter ureafaciens],4FFI_B Crystal Structure of Levan Fructotransferase D54N mutant from Arthrobacter ureafaciens in complex with levanbiose [Paenarthrobacter ureafaciens],4FFI_C Crystal Structure of Levan Fructotransferase D54N mutant from Arthrobacter ureafaciens in complex with levanbiose [Paenarthrobacter ureafaciens],4FFI_D Crystal Structure of Levan Fructotransferase D54N mutant from Arthrobacter ureafaciens in complex with levanbiose [Paenarthrobacter ureafaciens] |
|
| 1.37e-61 | 23 | 506 | 8 | 501 | Crystal structure of exo-inulinase from Aspergillus awamori in spacegroup P21 [Aspergillus awamori],1Y9G_A Crystal structure of exo-inulinase from Aspergillus awamori complexed with fructose [Aspergillus awamori],1Y9M_A Crystal structure of exo-inulinase from Aspergillus awamori in spacegroup P212121 [Aspergillus awamori] |
|
| 2.86e-44 | 23 | 338 | 8 | 316 | Structure of Saccharomyces cerevisiae invertase [Saccharomyces cerevisiae S288C],4EQV_B Structure of Saccharomyces cerevisiae invertase [Saccharomyces cerevisiae S288C],4EQV_C Structure of Saccharomyces cerevisiae invertase [Saccharomyces cerevisiae S288C],4EQV_D Structure of Saccharomyces cerevisiae invertase [Saccharomyces cerevisiae S288C],4EQV_E Structure of Saccharomyces cerevisiae invertase [Saccharomyces cerevisiae S288C],4EQV_F Structure of Saccharomyces cerevisiae invertase [Saccharomyces cerevisiae S288C],4EQV_G Structure of Saccharomyces cerevisiae invertase [Saccharomyces cerevisiae S288C],4EQV_H Structure of Saccharomyces cerevisiae invertase [Saccharomyces cerevisiae S288C] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 3.40e-96 | 23 | 506 | 44 | 499 | Levanbiose-producing levanase OS=Bacillus subtilis (strain 168) OX=224308 GN=levB PE=1 SV=1 |
|
| 2.68e-87 | 37 | 350 | 1 | 300 | Levanbiose-producing levanase (Fragment) OS=Geobacillus stearothermophilus OX=1422 GN=levB PE=1 SV=2 |
|
| 4.27e-76 | 23 | 506 | 35 | 497 | Levanase OS=Bacillus subtilis (strain 168) OX=224308 GN=sacC PE=1 SV=1 |
|
| 1.03e-61 | 23 | 506 | 27 | 520 | Extracellular exo-inulinase inuE OS=Aspergillus niger (strain CBS 513.88 / FGSC A1513) OX=425011 GN=inuE PE=2 SV=1 |
|
| 5.43e-61 | 23 | 506 | 27 | 520 | Extracellular exo-inulinase inuE OS=Aspergillus niger OX=5061 GN=inuE PE=1 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as SP
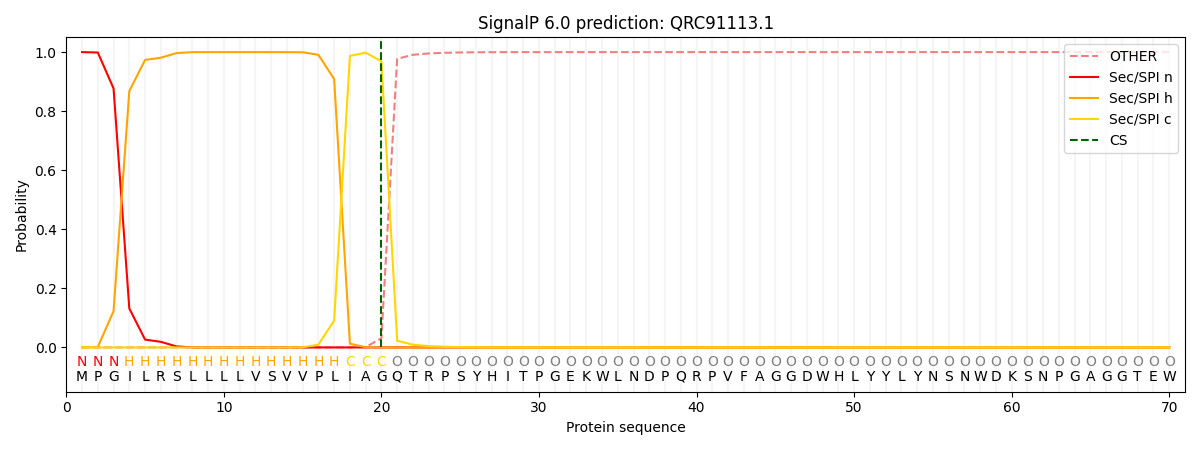
| Other | SP_Sec_SPI | CS Position |
|---|---|---|
| 0.000206 | 0.999752 | CS pos: 20-21. Pr: 0.9691 |
