You are browsing environment: FUNGIDB
CAZyme Information: PADG_00407-t30_1-p1
You are here: Home > Sequence: PADG_00407-t30_1-p1
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Paracoccidioides brasiliensis | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Ascomycota; Eurotiomycetes; ; NA; Paracoccidioides; Paracoccidioides brasiliensis | |||||||||||
| CAZyme ID | PADG_00407-t30_1-p1 | |||||||||||
| CAZy Family | AA3 | |||||||||||
| CAZyme Description | N-acetylglucosamine-6-phosphate deacetylase | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | ||||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| CE9 | 16 | 455 | 5.2e-111 | 0.9919571045576407 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| 238434 | NagA | 1.84e-121 | 14 | 457 | 1 | 374 | N-acetylglucosamine-6-phosphate deacetylase, NagA, catalyzes the hydrolysis of the N-acetyl group of N-acetyl-glucosamine-6-phosphate (GlcNAc-6-P) to glucosamine 6-phosphate and acetate. This is the first committed step in the biosynthetic pathway to amino-sugar-nucleotides, which is needed for cell wall peptidoglycan and teichoic acid biosynthesis. Deacetylation of N-acetylglucosamine is also important in lipopolysaccharide synthesis and cell wall recycling. |
| 224733 | NagA | 1.07e-91 | 55 | 462 | 39 | 380 | N-acetylglucosamine-6-phosphate deacetylase [Carbohydrate transport and metabolism]. |
| 272968 | nagA | 4.07e-68 | 57 | 450 | 45 | 376 | N-acetylglucosamine-6-phosphate deacetylase. [Central intermediary metabolism, Amino sugars] |
| 183010 | nagA | 3.24e-49 | 16 | 443 | 4 | 366 | N-acetylglucosamine-6-phosphate deacetylase; Provisional |
| 182623 | PRK10657 | 5.43e-08 | 355 | 454 | 268 | 369 | isoaspartyl dipeptidase; Provisional |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| 7.36e-266 | 1 | 468 | 1 | 465 | |
| 7.36e-266 | 1 | 468 | 1 | 465 | |
| 8.34e-199 | 11 | 465 | 10 | 448 | |
| 5.04e-198 | 11 | 466 | 9 | 424 | |
| 5.04e-198 | 11 | 466 | 9 | 424 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 2.71e-81 | 12 | 442 | 11 | 387 | Chain A, N-acetylglucosamine-6-phosphate deacetylase [Homo sapiens],7NUT_B Chain B, N-acetylglucosamine-6-phosphate deacetylase [Homo sapiens],7NUU_A Chain A, N-acetylglucosamine-6-phosphate deacetylase [Homo sapiens],7NUU_B Chain B, N-acetylglucosamine-6-phosphate deacetylase [Homo sapiens] |
|
| 6.40e-40 | 17 | 450 | 8 | 373 | N-acetylglucosamine-6-phosphate deacetylase from Vibrio cholerae. [Vibrio cholerae],3EGJ_B N-acetylglucosamine-6-phosphate deacetylase from Vibrio cholerae. [Vibrio cholerae],3IV8_A N-acetylglucosamine-6-phosphate deacetylase from Vibrio cholerae complexed with fructose 6-phosphate [Vibrio cholerae],3IV8_B N-acetylglucosamine-6-phosphate deacetylase from Vibrio cholerae complexed with fructose 6-phosphate [Vibrio cholerae],3IV8_C N-acetylglucosamine-6-phosphate deacetylase from Vibrio cholerae complexed with fructose 6-phosphate [Vibrio cholerae],3IV8_D N-acetylglucosamine-6-phosphate deacetylase from Vibrio cholerae complexed with fructose 6-phosphate [Vibrio cholerae] |
|
| 1.66e-39 | 65 | 450 | 54 | 381 | The Three-dimensional structure of the N-Acetylglucosamine-6- phosphate deacetylase from Bacillus subtilis [Bacillus subtilis],2VHL_B The Three-dimensional structure of the N-Acetylglucosamine-6- phosphate deacetylase from Bacillus subtilis [Bacillus subtilis] |
|
| 1.89e-33 | 61 | 450 | 46 | 373 | Crystal Structure of the N-Acetylglucosamine-6-phosphate deacetylase from Escherichia coli K12 [Escherichia coli K-12],1YMY_B Crystal Structure of the N-Acetylglucosamine-6-phosphate deacetylase from Escherichia coli K12 [Escherichia coli K-12],1YRR_A Crystal Structure Of The N-Acetylglucosamine-6-Phosphate Deacetylase From Escherichia Coli K12 at 2.0 A Resolution [Escherichia coli],1YRR_B Crystal Structure Of The N-Acetylglucosamine-6-Phosphate Deacetylase From Escherichia Coli K12 at 2.0 A Resolution [Escherichia coli],2P50_A Crystal structure of N-acetyl-D-Glucosamine-6-Phosphate deacetylase liganded with Zn [Escherichia coli K-12],2P50_B Crystal structure of N-acetyl-D-Glucosamine-6-Phosphate deacetylase liganded with Zn [Escherichia coli K-12],2P50_C Crystal structure of N-acetyl-D-Glucosamine-6-Phosphate deacetylase liganded with Zn [Escherichia coli K-12],2P50_D Crystal structure of N-acetyl-D-Glucosamine-6-Phosphate deacetylase liganded with Zn [Escherichia coli K-12] |
|
| 9.34e-33 | 61 | 450 | 46 | 373 | Chain A, N-acetylglucosamine-6-phosphate deacetylase [Escherichia coli K-12],2P53_B Chain B, N-acetylglucosamine-6-phosphate deacetylase [Escherichia coli K-12] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 1.97e-82 | 13 | 461 | 12 | 402 | N-acetylglucosamine-6-phosphate deacetylase OS=Danio rerio OX=7955 GN=amdhd2 PE=2 SV=1 |
|
| 1.39e-80 | 12 | 442 | 11 | 387 | N-acetylglucosamine-6-phosphate deacetylase OS=Homo sapiens OX=9606 GN=AMDHD2 PE=1 SV=2 |
|
| 1.53e-79 | 13 | 467 | 12 | 408 | N-acetylglucosamine-6-phosphate deacetylase OS=Mus musculus OX=10090 GN=Amdhd2 PE=1 SV=1 |
|
| 4.29e-79 | 13 | 442 | 12 | 387 | N-acetylglucosamine-6-phosphate deacetylase OS=Bos taurus OX=9913 GN=AMDHD2 PE=2 SV=1 |
|
| 4.29e-79 | 13 | 467 | 12 | 408 | N-acetylglucosamine-6-phosphate deacetylase OS=Rattus norvegicus OX=10116 GN=Amdhd2 PE=3 SV=2 |
SignalP and Lipop Annotations help
This protein is predicted as OTHER
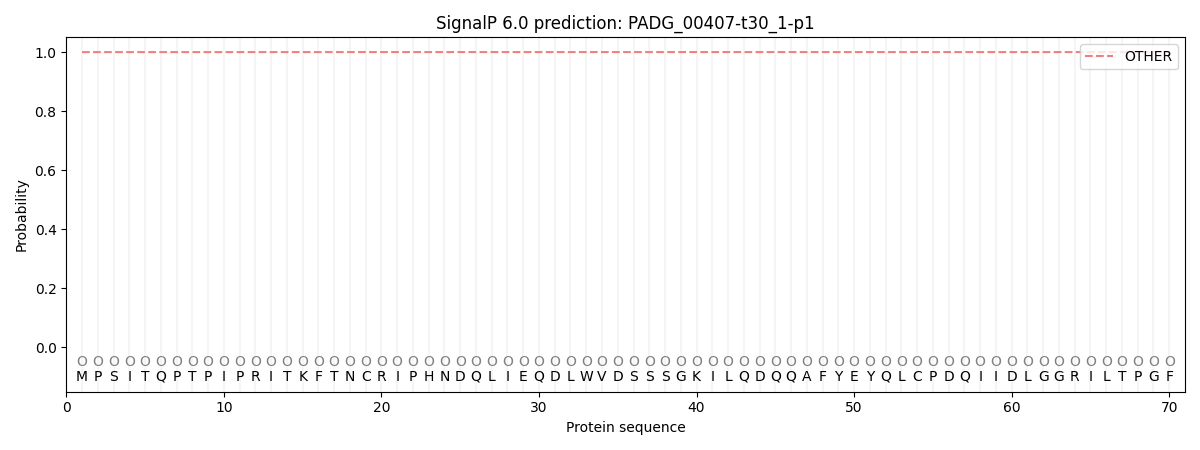
| Other | SP_Sec_SPI | CS Position |
|---|---|---|
| 1.000051 | 0.000003 |
