You are browsing environment: FUNGIDB
CAZyme Information: P174DRAFT_515722-t37_1-p1
You are here: Home > Sequence: P174DRAFT_515722-t37_1-p1
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
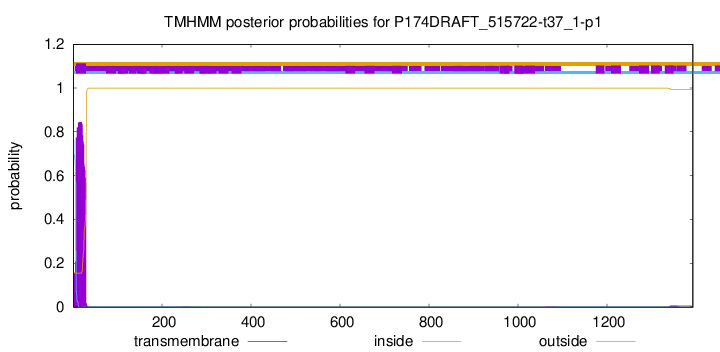
TMHMM annotations
Basic Information help
| Species | Aspergillus novofumigatus | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Ascomycota; Eurotiomycetes; ; Aspergillaceae; Aspergillus; Aspergillus novofumigatus | |||||||||||
| CAZyme ID | P174DRAFT_515722-t37_1-p1 | |||||||||||
| CAZy Family | GT71 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | ||||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH55 | 153 | 810 | 1.7e-163 | 0.9216216216216216 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| 176558 | PI-PLCXDc_like_2 | 1.33e-113 | 1002 | 1307 | 1 | 300 | Catalytic domain of uncharacterized hypothetical proteins similar to eukaryotic phosphatidylinositol-specific phospholipase C, X domain containing proteins. This subfamily corresponds to the catalytic domain present in a group of uncharacterized hypothetical proteins found in bacteria and fungi, which are similar to eukaryotic phosphatidylinositol-specific phospholipase C, X domain containing proteins (PI-PLCXD). The typical eukaryotic phosphoinositide-specific phospholipase C (PI-PLC, EC 3.1.4.11) has a multidomain organization that consists of a PLC catalytic core domain, and various regulatory domains. The catalytic core domain is assembled from two highly conserved X- and Y-regions split by a divergent linker sequence. In contrast, eukaryotic PI-PLCXDs contain a single TIM-barrel type catalytic domain, X domain, and are more closely related to bacterial PI-PLCs, which participate in Ca2+-independent PI metabolism, hydrolyzing the membrane lipid phosphatidylinositol (PI) to produce phosphorylated myo-inositol and diacylglycerol (DAG). Although the biological function of eukaryotic PI-PLCXDs still remains unclear, it may distinct from that of typical eukaryotic PI-PLCs. |
| 176529 | PI-PLCXDc_like | 5.90e-53 | 1002 | 1307 | 1 | 288 | Catalytic domain of phosphatidylinositol-specific phospholipase C X domain containing and similar proteins. This family corresponds to the catalytic domain present in phosphatidylinositol-specific phospholipase C X domain containing proteins (PI-PLCXD) which are bacterial phosphatidylinositol-specific phospholipase C (PI-PLC, EC 4.6.1.13) sequence homologs mainly found in eukaryota. The typical eukaryotic phosphoinositide-specific phospholipase C (PI-PLC, EC 3.1.4.11) have a multidomain organization that consists of a PLC catalytic core domain, and various regulatory domains. The catalytic core domain is assembled from two highly conserved X- and Y-regions split by a divergent linker sequence. In contrast, eukaryotic PI-PLCXDs and their bacterial homologs contain a single TIM-barrel type catalytic domain, X domain, which is more closely related to that of bacterial PI-PLCs. Although the biological function of eukaryotic PI-PLCXDs still remains unclear, it may be distinct from that of typical eukaryotic PI-PLCs. |
| 403800 | Pectate_lyase_3 | 2.24e-38 | 153 | 341 | 9 | 213 | Pectate lyase superfamily protein. This family of proteins possesses a beta helical structure like Pectate lyase. This family is most closely related to glycosyl hydrolase family 28. |
| 176500 | PI-PLCc_bacteria_like | 1.86e-30 | 1002 | 1283 | 1 | 259 | Catalytic domain of bacterial phosphatidylinositol-specific phospholipase C and similar proteins. This subfamily corresponds to the catalytic domain present in bacterial phosphatidylinositol-specific phospholipase C (PI-PLC, EC 4.6.1.13) and their sequence homologs found in eukaryota. Bacterial PI-PLCs participate in Ca2+-independent PI metabolism, hydrolyzing the membrane lipid phosphatidylinositol (PI) to produce phosphorylated myo-inositol and diacylglycerol (DAG). Although their precise physiological function remains unclear, bacterial PI-PLCs may function as virulence factors in some pathogenic bacteria. Bacterial PI-PLCs contain a single TIM-barrel type catalytic domain. Its catalytic mechanism is based on general base and acid catalysis utilizing two well conserved histidines, and consists of two steps, a phosphotransfer and a phosphodiesterase reaction. Eukaryotic homologs in this family are named as phosphatidylinositol-specific phospholipase C X domain containing proteins (PI-PLCXD). They are distinct from the typical eukaryotic phosphoinositide-specific phospholipases C (PI-PLC, EC 3.1.4.11), which have a multidomain organization that consists of a PLC catalytic core domain, and various regulatory domains. The catalytic core domain is assembled from two highly conserved X- and Y-regions split by a divergent linker sequence. In contrast, eukaryotic PI-PLCXDs contain a single TIM-barrel type catalytic domain, X domain, which is closely related to that of bacterial PI-PLCs. Although the biological function of eukaryotic PI-PLCXDs still remains unclear, it may be distinct from that of typical eukaryotic PI-PLCs. This family also includes a distinctly different type of eukaryotic PLC, glycosylphosphatidylinositol-specific phospholipase C (GPI-PLC), an integral membrane protein characterized in the protozoan parasite Trypanosoma brucei. T. brucei GPI-PLC hydrolyzes the GPI-anchor on the variant specific glycoprotein (VSG), releasing dimyristyl glycerol (DMG), which may facilitate the evasion of the protozoan to the host's immune system. It does not require Ca2+ for its activity and is more closely related to bacterial PI-PLCs, but not mammalian PI-PLCs. |
| 176555 | PI-PLCXD1c | 1.06e-08 | 1001 | 1077 | 1 | 94 | Catalytic domain of phosphatidylinositol-specific phospholipase C, X domain containing 1. This subfamily corresponds to the catalytic domain present in a group of phosphatidylinositol-specific phospholipase C X domain containing 1 (PI-PLCXD1), 2 (PI-PLCXD2) and 3 (PI-PLCXD3), which are bacterial phosphatidylinositol-specific phospholipase C (PI-PLC, EC 4.6.1.13) sequence homologs found in vertebrates. The typical eukaryotic phosphoinositide-specific phospholipase C (PI-PLC, EC 3.1.4.11) has a multidomain organization that consists of a PLC catalytic core domain, and various regulatory domains. The catalytic core domain is assembled from two highly conserved X- and Y-regions split by a divergent linker sequence. In contrast, members in this group contain a single TIM-barrel type catalytic domain, X domain, and are more closely related to bacterial PI-PLCs, which participate in Ca2+-independent PI metabolism, hydrolyzing the membrane lipid phosphatidylinositol (PI) to produce phosphorylated myo-inositol and diacylglycerol (DAG). Although the biological function of eukaryotic PI-PLCXDs still remains unclear, it may distinct from that of typical eukaryotic PI-PLCs. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| 7.25e-307 | 53 | 834 | 61 | 909 | |
| 1.16e-300 | 53 | 834 | 62 | 892 | |
| 5.87e-295 | 50 | 831 | 59 | 898 | |
| 2.96e-260 | 50 | 1320 | 63 | 1443 | |
| 3.33e-228 | 52 | 825 | 450 | 1296 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 2.76e-114 | 157 | 810 | 40 | 741 | Chain A, Beta-1,3-glucanase [Thermochaetoides thermophila],5M60_A Chain A, Beta-1,3-glucanase [Thermochaetoides thermophila] |
|
| 2.68e-102 | 140 | 781 | 26 | 716 | Chain A, Glucan 1,3-beta-glucosidase [Phanerodontia chrysosporium],3EQN_B Chain B, Glucan 1,3-beta-glucosidase [Phanerodontia chrysosporium],3EQO_A Chain A, Glucan 1,3-beta-glucosidase [Phanerodontia chrysosporium],3EQO_B Chain B, Glucan 1,3-beta-glucosidase [Phanerodontia chrysosporium] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 3.78e-107 | 122 | 770 | 25 | 824 | Probable glucan endo-1,3-beta-glucosidase ARB_02077 OS=Arthroderma benhamiae (strain ATCC MYA-4681 / CBS 112371) OX=663331 GN=ARB_02077 PE=1 SV=1 |
|
| 2.24e-85 | 157 | 763 | 82 | 721 | Glucan 1,3-beta-glucosidase OS=Cochliobolus carbonum OX=5017 GN=EXG1 PE=1 SV=1 |
|
| 2.36e-37 | 235 | 825 | 159 | 760 | Glucan endo-1,3-beta-glucosidase BGN13.1 OS=Trichoderma harzianum OX=5544 GN=bgn13.1 PE=1 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as SP

| Other | SP_Sec_SPI | CS Position |
|---|---|---|
| 0.000282 | 0.999726 | CS pos: 27-28. Pr: 0.9729 |