You are browsing environment: FUNGIDB
CAZyme Information: KNZ47113.1
You are here: Home > Sequence: KNZ47113.1
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Puccinia sorghi | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Basidiomycota; Pucciniomycetes; ; Pucciniaceae; Puccinia; Puccinia sorghi | |||||||||||
| CAZyme ID | KNZ47113.1 | |||||||||||
| CAZy Family | CE4 | |||||||||||
| CAZyme Description | NodB homology domain-containing protein [Source:UniProtKB/TrEMBL;Acc:A0A0L6UEW6] | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | ||||||||||||
Enzyme Prediction help
| EC | 3.5.1.41:1 | 3.5.1.41:1 |
|---|
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| CE4 | 82 | 230 | 1.1e-19 | 0.8846153846153846 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| 200575 | CE4_ClCDA_like | 4.22e-59 | 76 | 280 | 1 | 189 | Catalytic NodB homology domain of Colletotrichum lindemuthianum chitin deacetylase and similar proteins. This family is represented by the chitin deacetylase (endo-chitin de-N-acetylase, ClCDA, EC 3.5.1.41) from Colletotrichum lindemuthianum (also known as Glomerella lindemuthiana), which is a member of the carbohydrate esterase 4 (CE4) superfamily. ClCDA catalyzes the hydrolysis of N-acetamido groups of N-acetyl-D-glucosamine residues in chitin, converting it to chitosan in fungal cell walls. It consists of a single catalytic domain similar to the deformed (alpha/beta)8 barrel fold adopted by other CE4 esterases, which encompasses a mononuclear metalloenzyme employing a conserved His-His-Asp zinc-binding triad closely associated with the conserved catalytic base (aspartic acid) and acid (histidine), to carry out acid/base catalysis. It possesses a highly conserved substrate-binding groove, with subtle alterations that influence substrate specificity and subsite affinity. Unlike its bacterial homologs, ClCDA contains two intramolecular disulfide bonds that may add stability to this secreted protein. The family also includes many uncharacterized deacetylases and hypothetical proteins mainly from eukaryotes, which show high sequence similarity to ClCDA. |
| 213022 | CE4_NodB_like_6s_7s | 6.59e-37 | 83 | 281 | 1 | 166 | Catalytic NodB homology domain of rhizobial NodB-like proteins. This family belongs to the large and functionally diverse carbohydrate esterase 4 (CE4) superfamily, whose members show strong sequence similarity with some variability due to their distinct carbohydrate substrates. It includes many rhizobial NodB chitooligosaccharide N-deacetylase (EC 3.5.1.-)-like proteins, mainly from bacteria and eukaryotes, such as chitin deacetylases (EC 3.5.1.41), bacterial peptidoglycan N-acetylglucosamine deacetylases (EC 3.5.1.-), and acetylxylan esterases (EC 3.1.1.72), which catalyze the N- or O-deacetylation of substrates such as acetylated chitin, peptidoglycan, and acetylated xylan. All members of this family contain a catalytic NodB homology domain with the same overall topology and a deformed (beta/alpha)8 barrel fold with 6- or 7 strands. Their catalytic activity is dependent on the presence of a divalent cation, preferably cobalt or zinc, and they employ a conserved His-His-Asp zinc-binding triad closely associated with the conserved catalytic base (aspartic acid) and acid (histidine) to carry out acid/base catalysis. Several family members show diversity both in metal ion specificities and in the residues that coordinate the metal. |
| 396211 | Polysacc_deac_1 | 5.12e-28 | 83 | 230 | 7 | 122 | Polysaccharide deacetylase. This domain is found in polysaccharide deacetylase. This family of polysaccharide deacetylases includes NodB (nodulation protein B from Rhizobium) which is a chitooligosaccharide deacetylase. It also includes chitin deacetylase from yeast, and endoxylanases which hydrolyzes glucosidic bonds in xylan. |
| 200576 | CE4_MrCDA_like | 2.58e-24 | 83 | 282 | 1 | 175 | Catalytic NodB homology domain of Mucor rouxii chitin deacetylase and similar proteins. This family is represented by the chitin deacetylase (MrCDA, EC 3.5.1.41) encoded from the fungus Mucor rouxii (also known as Amylomyces rouxii). MrCDA is an acidic glycoprotein with a very stringent specificity for beta1-4-linked N-acetylglucosamine homopolymers. It requires at least four residues (chitotetraose) for catalysis, and can achieve extensive deacetylation on chitin polymers. MrCDA shows high sequence similarity to Colletotrichum lindemuthianum chitin deacetylase (endo-chitin de-N-acetylase, ClCDA), which consists of a single catalytic domain similar to the deformed (beta/alpha)8 barrel fold adopted by the carbohydrate esterase 4 (CE4) superfamily, which encompasses a mononuclear metalloenzyme employing a conserved His-His-Asp zinc-binding triad closely associated with the conserved catalytic base (aspartic acid) and acid (histidine) to carry out acid/base catalysis. The family also includes some uncharacterized eukaryotic and bacterial homologs of MrCDA. |
| 200569 | CE4_SmPgdA_like | 1.24e-22 | 87 | 271 | 5 | 164 | Catalytic NodB homology domain of Streptococcus mutans polysaccharide deacetylase PgdA, Bacillus subtilis YheN, and similar proteins. This family is represented by a putative polysaccharide deacetylase PgdA from the oral pathogen Streptococcus mutans (SmPgdA) and Bacillus subtilis YheN (BsYheN), which are members of the carbohydrate esterase 4 (CE4) superfamily. SmPgdA is an extracellular metal-dependent polysaccharide deacetylase with a typical CE4 fold, with metal bound to a His-His-Asp triad. It possesses de-N-acetylase activity toward a hexamer of chitooligosaccharide N-acetylglucosamine, but not shorter chitooligosaccharides or a synthetic peptidoglycan tetrasaccharide. SmPgdA plays a role in tuning cell surface properties and in interactions with (salivary) agglutinin, an essential component of the innate immune system, most likely through deacetylation of an as-yet-unidentified polysaccharide. SmPgdA shows significant homology to the catalytic domains of peptidoglycan deacetylases from Streptococcus pneumoniae (SpPgdA) and Listeria monocytogenes (LmPgdA), both of which are involved in the bacterial defense mechanism against human mucosal lysozyme. The Bacillus subtilis genome contains six polysaccharide deacetylase gene homologs: pdaA, pdaB (previously known as ybaN), yheN, yjeA, yxkH and ylxY. The biological function of BsYheN is still unknown. This family also includes many uncharacterized polysaccharide deacetylases mainly found in bacteria. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| 1.20e-60 | 64 | 332 | 22 | 253 | |
| 1.58e-54 | 69 | 278 | 40 | 220 | |
| 3.66e-47 | 86 | 338 | 47 | 266 | |
| 1.63e-42 | 59 | 332 | 15 | 249 | |
| 2.30e-42 | 73 | 332 | 29 | 249 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 1.02e-15 | 74 | 317 | 24 | 229 | Chain A, CHITIN DEACETYLASE [Aspergillus nidulans],2Y8U_B Chain B, CHITIN DEACETYLASE [Aspergillus nidulans] |
|
| 2.32e-15 | 79 | 281 | 21 | 186 | T48 deacetylase [Arthrobacter sp. AW19M34-1],5LGC_A T48 deacetylase with substrate [Arthrobacter sp. AW19M34-1] |
|
| 3.05e-10 | 86 | 318 | 45 | 247 | Structure of the chitin deacetylase from the fungal pathogen Colletotrichum lindemuthianum [Colletotrichum lindemuthianum] |
|
| 3.44e-09 | 83 | 236 | 60 | 182 | Structure of Bacillus subtilis PdaA in complex with NAG, a family 4 Carbohydrate esterase. [Bacillus subtilis],1W1A_2 Structure of Bacillus subtilis PdaA in complex with NAG, a family 4 Carbohydrate esterase. [Bacillus subtilis],1W1B_1 Structure of Bacillus subtilis PdaA with Cadmium, a family 4 Carbohydrate esterase. [Bacillus subtilis],1W1B_2 Structure of Bacillus subtilis PdaA with Cadmium, a family 4 Carbohydrate esterase. [Bacillus subtilis] |
|
| 3.62e-09 | 83 | 236 | 66 | 188 | Structure of Bacillus subtilis PdaA, a family 4 Carbohydrate esterase. [Bacillus subtilis],1W17_B Structure of Bacillus subtilis PdaA, a family 4 Carbohydrate esterase. [Bacillus subtilis] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 2.73e-17 | 75 | 207 | 31 | 140 | Chitin deacetylase OS=Pestalotiopsis sp. OX=36460 GN=CDA PE=1 SV=1 |
|
| 6.59e-16 | 75 | 317 | 160 | 367 | Chitin deacetylase OS=Arthroderma benhamiae (strain ATCC MYA-4681 / CBS 112371) OX=663331 GN=CDA PE=3 SV=1 |
|
| 5.88e-15 | 74 | 317 | 31 | 236 | Chitin deacetylase OS=Emericella nidulans (strain FGSC A4 / ATCC 38163 / CBS 112.46 / NRRL 194 / M139) OX=227321 GN=cda PE=1 SV=1 |
|
| 1.59e-10 | 74 | 317 | 140 | 348 | Probable peptidoglycan-N-acetylglucosamine deacetylase ARB_03699 OS=Arthroderma benhamiae (strain ATCC MYA-4681 / CBS 112371) OX=663331 GN=ARB_03699 PE=1 SV=2 |
|
| 1.48e-09 | 86 | 318 | 45 | 247 | Chitin deacetylase OS=Colletotrichum lindemuthianum OX=290576 GN=CDA PE=1 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as SP
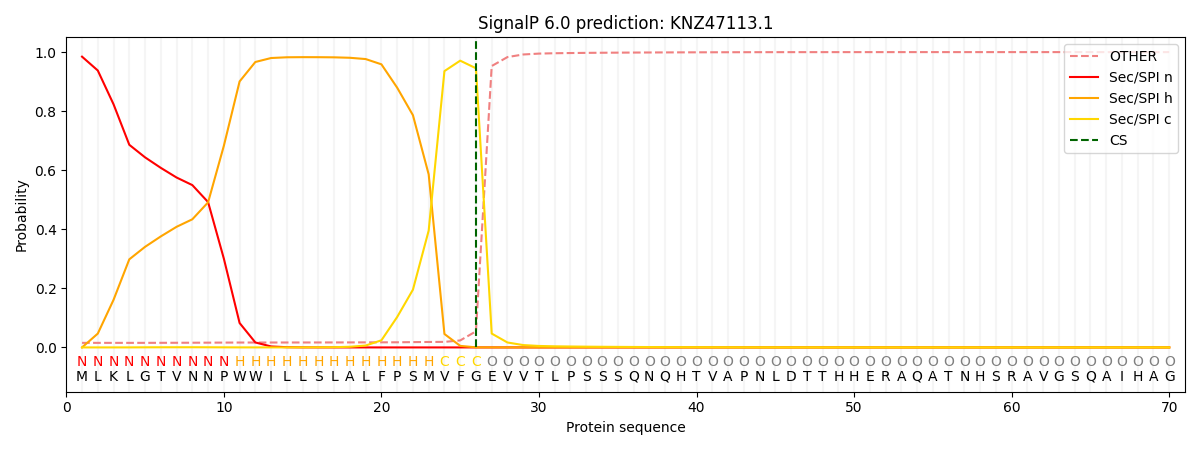
| Other | SP_Sec_SPI | CS Position |
|---|---|---|
| 0.017422 | 0.982560 | CS pos: 26-27. Pr: 0.9453 |
