You are browsing environment: FUNGIDB
CAZyme Information: KAF5687483.1
You are here: Home > Sequence: KAF5687483.1
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Fusarium circinatum | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Ascomycota; Sordariomycetes; ; Nectriaceae; Fusarium; Fusarium circinatum | |||||||||||
| CAZyme ID | KAF5687483.1 | |||||||||||
| CAZy Family | GT15 | |||||||||||
| CAZyme Description | laccase 2 | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | ||||||||||||
Enzyme Prediction help
| EC | 1.10.3.2:4 | 1.10.3.-:1 |
|---|
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| AA1 | 168 | 435 | 1.2e-27 | 0.38268156424581007 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| 259968 | CuRO_3_MaLCC_like | 3.11e-60 | 295 | 439 | 26 | 157 | The third cupredoxin domain of the fungal laccases similar to Ma-LCC from Melanocarpus albomyces. The subfamily of fungal laccases includes Ma-LCC and similar proteins. Ma-LCC is a multicopper oxidase (MCO) from Melanocarpus albomyces. Its crystal structure contains all four coppers at the mono- and trinuclear copper centers. Laccase is a blue multi-copper enzyme that catalyzes the oxidation of a variety aromatic - notably phenolic and inorganic substances coupled to the reduction of molecular oxygen to water. It has been implicated in a wide spectrum of biological activities and, in particular, plays a key role in morphogenesis, development and lignin metabolism in fungi and plants. Although MCOs have diverse functions, majority of them have three cupredoxin domain repeats that include one mononuclear and one trinuclear copper center. The copper ions are bound in several sites: Type 1, Type 2, and/or Type 3. The ensemble of types 2 and 3 copper is called a trinuclear cluster. MCOs oxidize their substrate by accepting electrons at a mononuclear copper center and transferring them to the active site trinuclear copper center. The cupredoxin domain 3 of 3-domain MCOs contains the Type 1 (T1) copper binding site and part the trinuclear copper binding site, which is located at the interface of domains 1 and 3. |
| 259923 | CuRO_1_MaLCC_like | 1.18e-33 | 69 | 154 | 1 | 85 | The first cupredoxin domain of the fungal laccases similar to Ma-LCC from Melanocarpus albomyces. The subfamily of fungal laccases includes Ma-LCC and similar proteins. Ma-LCC is a multicopper oxidase (MCO) from Melanocarpus albomyces. Its crystal structure contains all four coppers at the mono- and trinuclear copper centers. Laccase is a blue multi-copper enzyme that catalyzes the oxidation of a variety aromatic - notably phenolic and inorganic substances coupled to the reduction of molecular oxygen to water. It has been implicated in a wide spectrum of biological activities and, in particular, plays a key role in morphogenesis, development and lignin metabolism in fungi and plants. Although MCOs have diverse functions, majority of them have three cupredoxin domain repeats that include one mononuclear and one trinuclear copper center. The copper ions are bound in several sites: Type 1, Type 2, and/or Type 3. The ensemble of types 2 and 3 copper is called a trinuclear cluster. MCOs oxidize their substrate by accepting electrons at a mononuclear copper center and transferring them to the active site trinuclear copper center. The cupredoxin domain 1 of 3-domain MCOs contains part the trinuclear copper binding site, which is located at the interface of domains 1 and 3. |
| 400194 | Cu-oxidase_2 | 5.98e-32 | 320 | 444 | 29 | 138 | Multicopper oxidase. This entry contains many divergent copper oxidase-like domains that are not recognized by the pfam00394 model. |
| 259977 | CuRO_3_MCO_like_4 | 1.78e-29 | 315 | 440 | 50 | 166 | The third cupredoxin domain of uncharacterized multicopper oxidase. Multicopper Oxidases (MCOs) are multi-domain enzymes that are able to couple oxidation of substrates with reduction of dioxygen to water. MCOs oxidize their substrate by accepting electrons at a mononuclear copper centre and transferring them to a trinuclear copper centre which binds a dioxygen. The dioxygen, following the transfer of four electrons, is reduced to two molecules of water. These MCOs are capable of oxidizing a vast range of substrates, varying from aromatic to inorganic compounds such as metals. This subfamily of MCOs is composed of three cupredoxin domains. The cupredoxin domain 3 of 3-domain MCOs contains the Type 1 (T1) copper binding site and part the trinuclear copper binding site, which is located at the interface of domains 1 and 3. |
| 259972 | CuRO_3_tcLLC2_insect_like | 2.35e-28 | 357 | 436 | 68 | 161 | The third cupredoxin domain of the insect laccases similar to laccase 2 in Tribolium castaneum. This multicopper oxidase (MCO) family includes the majority of insect laccases. One member of the family is laccase 2 from Tribolium castaneum. Laccase 2 is required for beetle cuticle tanning. Laccase (polyphenol oxidase EC 1.10.3.2) is a blue multi-copper enzyme that catalyzes the oxidation of a variety of organic substrates coupled to the reduction of molecular oxygen to water. It displays broad substrate specificity, catalyzing the oxidation of a wide variety of aromatic - notably phenolic and inorganic substances. Laccase has been implicated in a wide spectrum of biological activities and, in particular, plays a key role in morphogenesis, development and lignin metabolism in fungi, plants and insects. Although MCOs have diverse functions, majority of them have three cupredoxin domain repeats that include one mononuclear and one trinuclear copper center. The copper ions are bound in several sites: Type 1, Type 2, and/or Type 3. The ensemble of types 2 and 3 copper is called a trinuclear cluster. MCOs oxidize their substrate by accepting electrons at a mononuclear copper center and transferring them to the active site trinuclear copper center. The cupredoxin domain 3 of 3-domain MCOs contains the Type 1 (T1) copper binding site and part the trinuclear copper binding site, which is located at the interface of domains 1 and 3. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| 0.0 | 24 | 487 | 62 | 659 | |
| 0.0 | 24 | 487 | 62 | 659 | |
| 0.0 | 24 | 487 | 62 | 659 | |
| 3.42e-301 | 24 | 487 | 62 | 659 | |
| 4.09e-221 | 21 | 487 | 54 | 651 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 5.02e-67 | 33 | 487 | 34 | 580 | Crystal structure of laccase from Botrytis aclada at 1.67 A resolution [Botrytis aclada],4X4K_A Structure of laccase from Botrytis aclada with full copper content [Botrytis aclada] |
|
| 9.80e-67 | 33 | 487 | 34 | 580 | Structure of the L499M mutant of the laccase from B.aclada [Botrytis aclada] |
|
| 1.79e-51 | 56 | 467 | 7 | 528 | Crystal structure of a laccase-like multicopper oxidase McoG from from Aspergillus niger [Aspergillus niger] |
|
| 1.82e-51 | 56 | 467 | 8 | 529 | Crystal structure of a laccase-like multicopper oxidase McoG from Aspergillus niger bound to zinc [Aspergillus niger] |
|
| 3.96e-51 | 56 | 467 | 35 | 556 | Crystal structure of the H253D mutant of McoG from Aspergillus niger [Aspergillus niger] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 8.32e-102 | 26 | 487 | 51 | 633 | Laccase ARB_05828 OS=Arthroderma benhamiae (strain ATCC MYA-4681 / CBS 112371) OX=663331 GN=ARB_05828 PE=1 SV=1 |
|
| 9.22e-66 | 29 | 487 | 28 | 561 | Laccase-1 OS=Botryotinia fuckeliana OX=40559 GN=lcc1 PE=2 SV=3 |
|
| 7.67e-64 | 37 | 487 | 38 | 581 | Laccase-2 OS=Botryotinia fuckeliana OX=40559 GN=lcc2 PE=2 SV=1 |
|
| 9.61e-62 | 34 | 487 | 40 | 590 | Oxidoreductase OpS5 OS=Beauveria bassiana (strain ARSEF 2860) OX=655819 GN=OpS5 PE=1 SV=1 |
|
| 5.36e-58 | 32 | 487 | 21 | 610 | Oxidoreductase ptaE OS=Pestalotiopsis fici (strain W106-1 / CGMCC3.15140) OX=1229662 GN=ptaE PE=2 SV=2 |
SignalP and Lipop Annotations help
This protein is predicted as OTHER
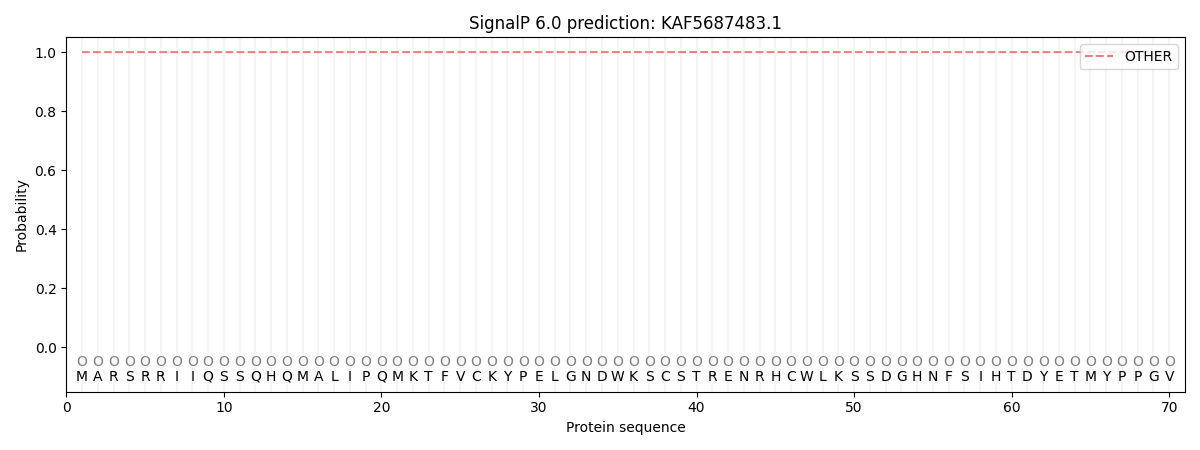
| Other | SP_Sec_SPI | CS Position |
|---|---|---|
| 1.000019 | 0.000028 |
