You are browsing environment: FUNGIDB
CAZyme Information: EEQ28605.1
You are here: Home > Sequence: EEQ28605.1
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Microsporum canis | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Ascomycota; Eurotiomycetes; ; Arthrodermataceae; Microsporum; Microsporum canis | |||||||||||
| CAZyme ID | EEQ28605.1 | |||||||||||
| CAZy Family | AA7 | |||||||||||
| CAZyme Description | FG-GAP repeat domain-containing protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | ||||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| CE3 | 418 | 597 | 4.4e-50 | 0.9536082474226805 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| 238871 | XynB_like | 3.47e-53 | 457 | 597 | 14 | 157 | SGNH_hydrolase subfamily, similar to Ruminococcus flavefaciens XynB. Most likely a secreted hydrolase with xylanase activity. SGNH hydrolases are a diverse family of lipases and esterases. The tertiary fold of the enzyme is substantially different from that of the alpha/beta hydrolase family and unique among all known hydrolases; its active site closely resembles the Ser-His-Asp(Glu) triad found in other serine hydrolases. |
| 404371 | Lipase_GDSL_2 | 2.02e-20 | 418 | 586 | 5 | 174 | GDSL-like Lipase/Acylhydrolase family. This family of presumed lipases and related enzymes are similar to pfam00657. |
| 238141 | SGNH_hydrolase | 3.34e-17 | 418 | 596 | 7 | 187 | SGNH_hydrolase, or GDSL_hydrolase, is a diverse family of lipases and esterases. The tertiary fold of the enzyme is substantially different from that of the alpha/beta hydrolase family and unique among all known hydrolases; its active site closely resembles the typical Ser-His-Asp(Glu) triad from other serine hydrolases, but may lack the carboxlic acid. |
| 199914 | M35_deuterolysin_like | 8.58e-15 | 57 | 299 | 1 | 167 | Peptidase M35 domain of deuterolysins and related proteins. This family M35 Zn2+-metallopeptidase extracellular domain is found in fungal deutrolysins (acid metalloproteinase, neutral proteinase II), including some well-characterized metallopeptidase domains in Aspergillus oryzae (NpII), Aspergillus fumigatus (MEP20), Penicillium roqueforti (protease II) and Emericella nidulans (PepJ peptidase). The neutral proteinase II from Aspergillus oryzae (NpII) unfolds reversibly upon incubation at higher temperatures, and loss in activity is mainly due to autoproteolysis. MEP20 is encoded by the mepB gene, which appears to be associated with the cytoplasmic degradation of small peptides. PepJ peptidase is a thermostable enzyme released under carbon starvation. Most members of this family contain a unique zinc-binding motif (the aspzincin motif), defined by the HExxH + D motif where an aspartic acid is the third zinc ligand and is found in a GTXDXXYG or similar motif C-terminal to the His zinc ligands. The aspzincin motif is poorly conserved in one subgroup, that includes Asp f2, a major allergen from Aspergillus fumigatus. This subgroup in addition lacks the key conserved Tyr residue which acts as a proton donor during catalysis, and no protease activity has been detected to date for Asp f2. |
| 239945 | SGNH_hydrolase_like_4 | 2.09e-09 | 418 | 592 | 9 | 178 | Members of the SGNH-hydrolase superfamily, a diverse family of lipases and esterases. The tertiary fold of the enzyme is substantially different from that of the alpha/beta hydrolase family and unique among all known hydrolases; its active site closely resembles the Ser-His-Asp(Glu) triad from other serine hydrolases, but may lack the carboxlic acid. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| 8.61e-113 | 399 | 935 | 21 | 498 | |
| 3.32e-93 | 398 | 946 | 312 | 824 | |
| 1.87e-91 | 397 | 946 | 263 | 769 | |
| 5.96e-91 | 399 | 935 | 57 | 501 | |
| 1.58e-89 | 399 | 935 | 144 | 588 |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 5.90e-07 | 479 | 614 | 23 | 150 | Putative endoglucanase X (Fragment) OS=Acetivibrio thermocellus OX=1515 GN=celX PE=1 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as SP
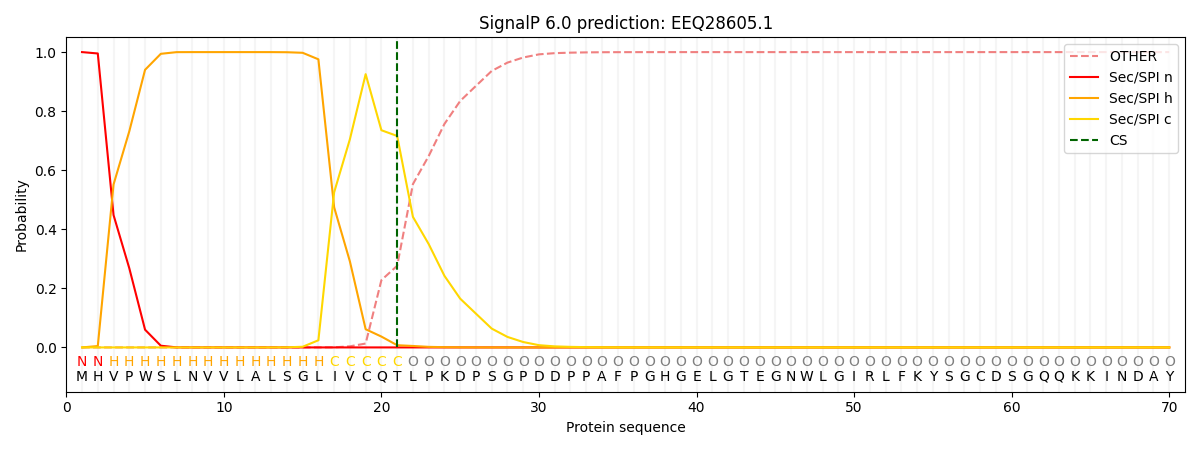
| Other | SP_Sec_SPI | CS Position |
|---|---|---|
| 0.000248 | 0.999738 | CS pos: 21-22. Pr: 0.7160 |
