You are browsing environment: FUNGIDB
CAZyme Information: CVL07594.1
You are here: Home > Sequence: CVL07594.1
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Fusarium mangiferae | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Ascomycota; Sordariomycetes; ; Nectriaceae; Fusarium; Fusarium mangiferae | |||||||||||
| CAZyme ID | CVL07594.1 | |||||||||||
| CAZy Family | GT2|GT2 | |||||||||||
| CAZyme Description | Related to Sucrose-6-phosphate hydrolase [Source:UniProtKB/TrEMBL;Acc:A0A1L7UA44] | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | ||||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH32 | 68 | 419 | 6.8e-59 | 0.962457337883959 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| 350133 | GH32_XdINV-like | 5.71e-148 | 74 | 418 | 1 | 335 | glycoside hydrolase family 32 protein such as Xanthophyllomyces dendrorhous beta-fructofuranosidase (Inv;Xd-INV;XdINV). This subfamily of glycosyl hydrolase family GH32 includes fructan:fructan 1-fructosyltransferase (FT, EC 2.4.1.100) and beta-fructofuranosidase (invertase or Inv, EC 3.2.1.26), among others. These enzymes cleave sucrose into fructose and glucose via beta-fructofuranosidase activity, producing invert sugar that is a mixture of dextrorotatory D-glucose and levorotatory D-fructose, thus named invertase (EC 3.2.1.26). These retaining enzymes (i.e. they retain the configuration at anomeric carbon atom of the substrate) catalyze hydrolysis in two steps involving a covalent glycosyl enzyme intermediate: an aspartate located close to the N-terminus acts as the catalytic nucleophile and a glutamate acts as the general acid/base; a conserved aspartate residue in the Arg-Asp-Pro (RDP) motif stabilizes the transition state. Xanthophyllomyces dendrorhous beta-fructofuranosidase (XdINV) also catalyzes the synthesis of fructooligosaccharides (FOS, a beneficial prebiotic), producing neo-FOS, making it an interesting biotechnology target. Structural studies show plasticity of its active site, having a flexible loop that is essential in binding sucrose and beta(2-1)-linked oligosaccharide, making it a valuable biocatalyst to produce novel bioconjugates. The breakdown of sucrose is widely used as a carbon or energy source by bacteria, fungi, and plants. Invertase is used commercially in the confectionery industry, since fructose has a sweeter taste than sucrose and a lower tendency to crystallize. A common structural feature of all these enzymes is a 5-bladed beta-propeller domain, similar to GH43, that contains the catalytic acid and catalytic base. A long V-shaped groove, partially enclosed at one end, forms a single extended substrate-binding surface across the face of the propeller. |
| 214757 | Glyco_32 | 1.60e-50 | 68 | 608 | 1 | 435 | Glycosyl hydrolases family 32. |
| 350110 | GH32_FFase | 7.12e-49 | 74 | 418 | 1 | 281 | Glycosyl hydrolase family 32, beta-fructosidases. Glycosyl hydrolase family GH32 cleaves sucrose into fructose and glucose via beta-fructofuranosidase activity, producing invert sugar that is a mixture of dextrorotatory D-glucose and levorotatory D-fructose, thus named invertase (EC 3.2.1.26). This family also contains other fructofuranosidases such as inulinase (EC 3.2.1.7), exo-inulinase (EC 3.2.1.80), levanase (EC 3.2.1.65), and transfructosidases such sucrose:sucrose 1-fructosyltransferase (EC 2.4.1.99), fructan:fructan 1-fructosyltransferase (EC 2.4.1.100), sucrose:fructan 6-fructosyltransferase (EC 2.4.1.10), fructan:fructan 6G-fructosyltransferase (EC 2.4.1.243) and levan fructosyltransferases (EC 2.4.1.-). These retaining enzymes (i.e. they retain the configuration at anomeric carbon atom of the substrate) catalyze hydrolysis in two steps involving a covalent glycosyl enzyme intermediate: an aspartate located close to the N-terminus acts as the catalytic nucleophile and a glutamate acts as the general acid/base; a conserved aspartate residue in the Arg-Asp-Pro (RDP) motif stabilizes the transition state. These enzymes are predicted to display a 5-fold beta-propeller fold as found for GH43 and CH68. The breakdown of sucrose is widely used as a carbon or energy source by bacteria, fungi, and plants. Invertase is used commercially in the confectionery industry, since fructose has a sweeter taste than sucrose and a lower tendency to crystallize. A common structural feature of all these enzymes is a 5-bladed beta-propeller domain, similar to GH43, that contains the catalytic acid and catalytic base. A long V-shaped groove, partially enclosed at one end, forms a single extended substrate-binding surface across the face of the propeller. |
| 395193 | Glyco_hydro_32N | 1.91e-38 | 68 | 418 | 1 | 297 | Glycosyl hydrolases family 32 N-terminal domain. This domain corresponds to the N-terminal domain of glycosyl hydrolase family 32 which forms a five bladed beta propeller structure. |
| 224536 | SacC | 3.16e-35 | 63 | 639 | 28 | 482 | Sucrose-6-phosphate hydrolase SacC, GH32 family [Carbohydrate transport and metabolism]. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| 0.0 | 1 | 658 | 1 | 658 | |
| 0.0 | 1 | 658 | 1 | 656 | |
| 0.0 | 1 | 658 | 1 | 656 | |
| 0.0 | 1 | 658 | 1 | 656 | |
| 0.0 | 1 | 599 | 1 | 607 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 4.53e-38 | 63 | 617 | 26 | 565 | Aspergillus kawachii beta-fructofuranosidase complexed with glycerol [Aspergillus luchuensis IFO 4308],5XH9_A Aspergillus kawachii beta-fructofuranosidase [Aspergillus luchuensis IFO 4308],5XHA_A Aspergillus kawachii beta-fructofuranosidase complexed with fructose [Aspergillus luchuensis IFO 4308] |
|
| 4.84e-36 | 63 | 619 | 65 | 602 | Chain A, Beta-fructofuranosidase [Phaffia rhodozyma],5ANN_B Chain B, Beta-fructofuranosidase [Phaffia rhodozyma] |
|
| 4.84e-36 | 63 | 619 | 65 | 602 | Chain A, Beta-fructofuranosidase [Phaffia rhodozyma],6S82_B Chain B, Beta-fructofuranosidase [Phaffia rhodozyma] |
|
| 5.05e-35 | 63 | 619 | 63 | 600 | Chain A, Beta-fructofuranosidase [Phaffia rhodozyma],5FK7_B Chain B, Beta-fructofuranosidase [Phaffia rhodozyma],5FK8_A Chain A, Beta-fructofuranosidase [Phaffia rhodozyma],5FK8_B Chain B, Beta-fructofuranosidase [Phaffia rhodozyma],5FKB_A Chain A, BETA-FRUCTOFURANOSIDASE [Phaffia rhodozyma],5FKB_B Chain B, BETA-FRUCTOFURANOSIDASE [Phaffia rhodozyma],5FKC_A Chain A, BETA-FRUCTOFURANOSIDASE [Phaffia rhodozyma],5FKC_B Chain B, BETA-FRUCTOFURANOSIDASE [Phaffia rhodozyma],5FMC_A Chain A, Beta-fructofuranosidase [Phaffia rhodozyma],5FMC_B Chain B, Beta-fructofuranosidase [Phaffia rhodozyma] |
|
| 5.11e-35 | 63 | 619 | 65 | 602 | Chain A, Beta-fructofuranosidase [Phaffia rhodozyma],5FIX_B Chain B, Beta-fructofuranosidase [Phaffia rhodozyma],5FMB_A Chain A, BETA-FRUCTOFURANOSIDASE [Phaffia rhodozyma],5FMB_B Chain B, BETA-FRUCTOFURANOSIDASE [Phaffia rhodozyma],5FMD_A Chain A, Beta-fructofuranosidase [Phaffia rhodozyma],5FMD_B Chain B, Beta-fructofuranosidase [Phaffia rhodozyma] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 8.26e-28 | 50 | 654 | 38 | 581 | Beta-fructofuranosidase, insoluble isoenzyme CWINV1 OS=Arabidopsis thaliana OX=3702 GN=CWINV1 PE=1 SV=1 |
|
| 8.45e-27 | 64 | 653 | 47 | 579 | Beta-fructofuranosidase, insoluble isoenzyme 3 OS=Oryza sativa subsp. japonica OX=39947 GN=CIN3 PE=2 SV=1 |
|
| 8.45e-27 | 64 | 653 | 47 | 579 | Beta-fructofuranosidase, insoluble isoenzyme 3 OS=Oryza sativa subsp. indica OX=39946 GN=CIN3 PE=2 SV=2 |
|
| 2.73e-26 | 54 | 652 | 38 | 580 | Beta-fructofuranosidase, insoluble isoenzyme CWINV2 OS=Arabidopsis thaliana OX=3702 GN=CWINV2 PE=2 SV=1 |
|
| 6.48e-26 | 64 | 658 | 54 | 589 | Beta-fructofuranosidase, cell wall isozyme OS=Zea mays OX=4577 PE=2 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as OTHER
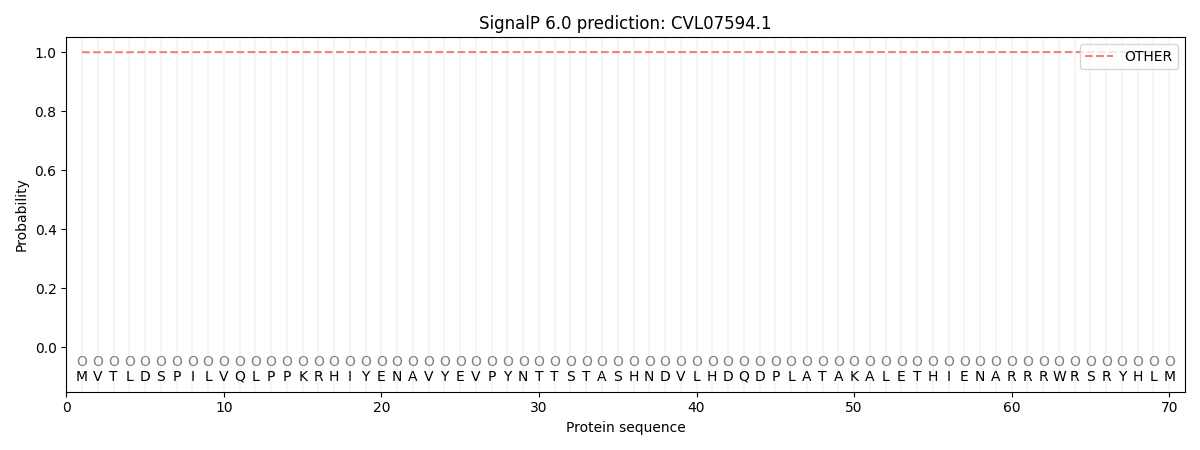
| Other | SP_Sec_SPI | CS Position |
|---|---|---|
| 0.999629 | 0.000395 |
