You are browsing environment: FUNGIDB
CAZyme Information: Afu5g13830-T-p1
You are here: Home > Sequence: Afu5g13830-T-p1
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
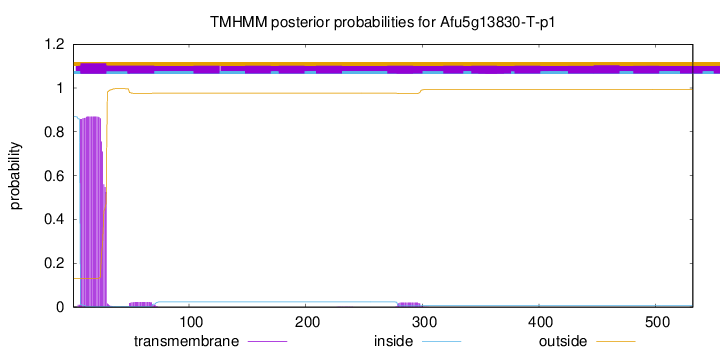
TMHMM annotations
Basic Information help
| Species | Aspergillus fumigatus | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Ascomycota; Eurotiomycetes; ; Aspergillaceae; Aspergillus; Aspergillus fumigatus | |||||||||||
| CAZyme ID | Afu5g13830-T-p1 | |||||||||||
| CAZy Family | GH5 | |||||||||||
| CAZyme Description | Putative alpha-galactosidase | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | ||||||||||||
Enzyme Prediction help
| EC | 3.2.1.49:3 | 3.2.1.22:2 | 3.2.1.49:2 | 3.2.1.22:2 |
|---|
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH27 | 126 | 381 | 3.6e-52 | 0.9781659388646288 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| 269893 | GH27 | 7.50e-107 | 32 | 317 | 1 | 271 | glycosyl hydrolase family 27 (GH27). GH27 enzymes occur in eukaryotes, prokaryotes, and archaea with a wide range of hydrolytic activities, including alpha-glucosidase (glucoamylase and sucrase-isomaltase), alpha-N-acetylgalactosaminidase, and 3-alpha-isomalto-dextranase. All GH27 enzymes cleave a terminal carbohydrate moiety from a substrate that varies considerably in size, depending on the enzyme, and may be either a starch or a glycoprotein. GH27 members are retaining enzymes that cleave their substrates via an acid/base-catalyzed, double-displacement mechanism involving a covalent glycosyl-enzyme intermediate. Two aspartic acid residues have been identified as the catalytic nucleophile and the acid/base, respectively. |
| 374582 | Melibiase_2 | 2.10e-78 | 31 | 317 | 1 | 284 | Alpha galactosidase A. |
| 166449 | PLN02808 | 1.36e-71 | 31 | 402 | 31 | 381 | alpha-galactosidase |
| 178295 | PLN02692 | 9.99e-69 | 31 | 402 | 55 | 406 | alpha-galactosidase |
| 177874 | PLN02229 | 1.91e-63 | 31 | 402 | 62 | 415 | alpha-galactosidase |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| 0.0 | 1 | 532 | 1 | 532 | |
| 1.79e-261 | 21 | 529 | 31 | 544 | |
| 1.27e-259 | 4 | 529 | 5 | 536 | |
| 8.82e-243 | 34 | 507 | 1 | 478 | |
| 1.96e-241 | 22 | 529 | 25 | 532 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 1.32e-58 | 31 | 402 | 8 | 357 | Chain A, alpha-galactosidase [Oryza sativa] |
|
| 1.43e-57 | 28 | 363 | 96 | 432 | Crystal structure of a putative alpha-galactosidase/melibiase (BF4189) from Bacteroides fragilis NCTC 9343 at 2.00 A resolution [Bacteroides fragilis NCTC 9343],4OGZ_B Crystal structure of a putative alpha-galactosidase/melibiase (BF4189) from Bacteroides fragilis NCTC 9343 at 2.00 A resolution [Bacteroides fragilis NCTC 9343] |
|
| 2.97e-57 | 28 | 383 | 5 | 360 | The Structure of alpha-N-Acetylgalactosaminidase [Gallus gallus],1KTC_A The Structure of alpha-N-Acetylgalactosaminidase [Gallus gallus] |
|
| 4.04e-57 | 28 | 371 | 5 | 347 | Chain A, Alpha-galactosidase A [Homo sapiens],3LX9_B Chain B, Alpha-galactosidase A [Homo sapiens],3LXA_A Chain A, Alpha-galactosidase A [Homo sapiens],3LXA_B Chain B, Alpha-galactosidase A [Homo sapiens],3LXB_A Chain A, Alpha-galactosidase A [Homo sapiens],3LXB_B Chain B, Alpha-galactosidase A [Homo sapiens],3LXC_A Chain A, Alpha-galactosidase A [Homo sapiens],3LXC_B Chain B, Alpha-galactosidase A [Homo sapiens] |
|
| 7.02e-56 | 28 | 371 | 5 | 347 | Structure of human alpha-galactosidase [Homo sapiens],1R46_B Structure of human alpha-galactosidase [Homo sapiens],1R47_A Structure of human alpha-galactosidase [Homo sapiens],1R47_B Structure of human alpha-galactosidase [Homo sapiens],3GXN_A Crystal structure of apo alpha-galactosidase A at pH 4.5 [Homo sapiens],3GXN_B Crystal structure of apo alpha-galactosidase A at pH 4.5 [Homo sapiens],3GXP_A Crystal structure of acid-alpha-galactosidase A complexed with galactose at pH 4.5 [Homo sapiens],3GXP_B Crystal structure of acid-alpha-galactosidase A complexed with galactose at pH 4.5 [Homo sapiens],3GXT_A Crystal structure of alpha-galactosidase A at pH 4.5 complexed with 1-deoxygalactonijirimycin [Homo sapiens],3GXT_B Crystal structure of alpha-galactosidase A at pH 4.5 complexed with 1-deoxygalactonijirimycin [Homo sapiens],3HG2_A Human alpha-galactosidase catalytic mechanism 1. Empty active site [Homo sapiens],3HG2_B Human alpha-galactosidase catalytic mechanism 1. Empty active site [Homo sapiens],3HG4_A Human alpha-galactosidase catalytic mechanism 3. Covalent intermediate [Homo sapiens],3HG4_B Human alpha-galactosidase catalytic mechanism 3. Covalent intermediate [Homo sapiens],3HG5_A Human alpha-galactosidase catalytic mechanism 4. Product bound [Homo sapiens],3HG5_B Human alpha-galactosidase catalytic mechanism 4. Product bound [Homo sapiens],3S5Y_A Pharmacological Chaperoning in Human alpha-Galactosidase [Homo sapiens],3S5Y_B Pharmacological Chaperoning in Human alpha-Galactosidase [Homo sapiens],3S5Z_A Pharmacological Chaperoning in Human alpha-Galactosidase [Homo sapiens],3S5Z_B Pharmacological Chaperoning in Human alpha-Galactosidase [Homo sapiens],4NXS_A Crystal structure of human alpha-galactosidase A in complex with 1-deoxygalactonojirimycin-pFPhT [Homo sapiens],4NXS_B Crystal structure of human alpha-galactosidase A in complex with 1-deoxygalactonojirimycin-pFPhT [Homo sapiens],6IBK_A Crystal structure of human alpha-galactosidase A in complex with alpha-galactose configured cyclosulfamidate ME763 [Homo sapiens],6IBK_B Crystal structure of human alpha-galactosidase A in complex with alpha-galactose configured cyclosulfamidate ME763 [Homo sapiens],6IBM_A Crystal structure of human alpha-galactosidase A in complex with alpha-galactose configured cyclosulfate ME776 [Homo sapiens],6IBM_B Crystal structure of human alpha-galactosidase A in complex with alpha-galactose configured cyclosulfate ME776 [Homo sapiens],6IBR_A Crystal structure of human alpha-galactosidase A in complex with alpha-galactose configured cyclophellitol epoxide LWA481 [Homo sapiens],6IBR_B Crystal structure of human alpha-galactosidase A in complex with alpha-galactose configured cyclophellitol epoxide LWA481 [Homo sapiens],6IBT_A Crystal structure of human alpha-galactosidase A in complex with alpha-galactose configured cyclophellitol aziridine ME737 [Homo sapiens],6IBT_B Crystal structure of human alpha-galactosidase A in complex with alpha-galactose configured cyclophellitol aziridine ME737 [Homo sapiens] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 0.0 | 1 | 532 | 1 | 532 | Probable alpha-galactosidase A OS=Neosartorya fischeri (strain ATCC 1020 / DSM 3700 / CBS 544.65 / FGSC A1164 / JCM 1740 / NRRL 181 / WB 181) OX=331117 GN=aglA PE=3 SV=1 |
|
| 0.0 | 1 | 532 | 1 | 532 | Probable alpha-galactosidase A OS=Neosartorya fumigata (strain ATCC MYA-4609 / Af293 / CBS 101355 / FGSC A1100) OX=330879 GN=aglA PE=3 SV=1 |
|
| 1.05e-281 | 10 | 530 | 6 | 524 | Probable alpha-galactosidase A OS=Aspergillus clavatus (strain ATCC 1007 / CBS 513.65 / DSM 816 / NCTC 3887 / NRRL 1 / QM 1276 / 107) OX=344612 GN=aglA PE=3 SV=1 |
|
| 3.18e-262 | 21 | 529 | 31 | 544 | Alpha-galactosidase A OS=Aspergillus niger OX=5061 GN=aglA PE=1 SV=1 |
|
| 2.25e-260 | 4 | 529 | 5 | 536 | Probable alpha-galactosidase A OS=Aspergillus niger (strain CBS 513.88 / FGSC A1513) OX=425011 GN=aglA PE=3 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as SP
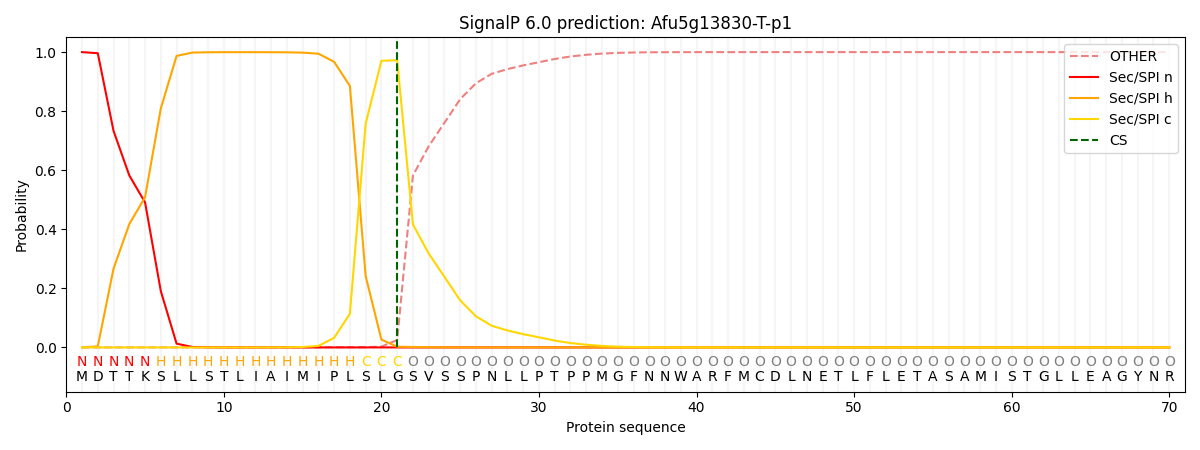
| Other | SP_Sec_SPI | CS Position |
|---|---|---|
| 0.001469 | 0.998512 | CS pos: 21-22. Pr: 0.9725 |