You are browsing environment: FUNGIDB
CAZyme Information: ATEG_04996-t26_1-p1
You are here: Home > Sequence: ATEG_04996-t26_1-p1
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Aspergillus terreus | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Ascomycota; Eurotiomycetes; ; Aspergillaceae; Aspergillus; Aspergillus terreus | |||||||||||
| CAZyme ID | ATEG_04996-t26_1-p1 | |||||||||||
| CAZy Family | GH25 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | ||||||||||||
Enzyme Prediction help
| EC | 3.2.1.26:10 | 2.4.1.100:6 | 2.4.1.-:2 |
|---|
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH32 | 47 | 398 | 1.2e-39 | 0.962457337883959 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| 350133 | GH32_XdINV-like | 4.83e-127 | 53 | 398 | 1 | 336 | glycoside hydrolase family 32 protein such as Xanthophyllomyces dendrorhous beta-fructofuranosidase (Inv;Xd-INV;XdINV). This subfamily of glycosyl hydrolase family GH32 includes fructan:fructan 1-fructosyltransferase (FT, EC 2.4.1.100) and beta-fructofuranosidase (invertase or Inv, EC 3.2.1.26), among others. These enzymes cleave sucrose into fructose and glucose via beta-fructofuranosidase activity, producing invert sugar that is a mixture of dextrorotatory D-glucose and levorotatory D-fructose, thus named invertase (EC 3.2.1.26). These retaining enzymes (i.e. they retain the configuration at anomeric carbon atom of the substrate) catalyze hydrolysis in two steps involving a covalent glycosyl enzyme intermediate: an aspartate located close to the N-terminus acts as the catalytic nucleophile and a glutamate acts as the general acid/base; a conserved aspartate residue in the Arg-Asp-Pro (RDP) motif stabilizes the transition state. Xanthophyllomyces dendrorhous beta-fructofuranosidase (XdINV) also catalyzes the synthesis of fructooligosaccharides (FOS, a beneficial prebiotic), producing neo-FOS, making it an interesting biotechnology target. Structural studies show plasticity of its active site, having a flexible loop that is essential in binding sucrose and beta(2-1)-linked oligosaccharide, making it a valuable biocatalyst to produce novel bioconjugates. The breakdown of sucrose is widely used as a carbon or energy source by bacteria, fungi, and plants. Invertase is used commercially in the confectionery industry, since fructose has a sweeter taste than sucrose and a lower tendency to crystallize. A common structural feature of all these enzymes is a 5-bladed beta-propeller domain, similar to GH43, that contains the catalytic acid and catalytic base. A long V-shaped groove, partially enclosed at one end, forms a single extended substrate-binding surface across the face of the propeller. |
| 350110 | GH32_FFase | 2.56e-37 | 53 | 397 | 1 | 281 | Glycosyl hydrolase family 32, beta-fructosidases. Glycosyl hydrolase family GH32 cleaves sucrose into fructose and glucose via beta-fructofuranosidase activity, producing invert sugar that is a mixture of dextrorotatory D-glucose and levorotatory D-fructose, thus named invertase (EC 3.2.1.26). This family also contains other fructofuranosidases such as inulinase (EC 3.2.1.7), exo-inulinase (EC 3.2.1.80), levanase (EC 3.2.1.65), and transfructosidases such sucrose:sucrose 1-fructosyltransferase (EC 2.4.1.99), fructan:fructan 1-fructosyltransferase (EC 2.4.1.100), sucrose:fructan 6-fructosyltransferase (EC 2.4.1.10), fructan:fructan 6G-fructosyltransferase (EC 2.4.1.243) and levan fructosyltransferases (EC 2.4.1.-). These retaining enzymes (i.e. they retain the configuration at anomeric carbon atom of the substrate) catalyze hydrolysis in two steps involving a covalent glycosyl enzyme intermediate: an aspartate located close to the N-terminus acts as the catalytic nucleophile and a glutamate acts as the general acid/base; a conserved aspartate residue in the Arg-Asp-Pro (RDP) motif stabilizes the transition state. These enzymes are predicted to display a 5-fold beta-propeller fold as found for GH43 and CH68. The breakdown of sucrose is widely used as a carbon or energy source by bacteria, fungi, and plants. Invertase is used commercially in the confectionery industry, since fructose has a sweeter taste than sucrose and a lower tendency to crystallize. A common structural feature of all these enzymes is a 5-bladed beta-propeller domain, similar to GH43, that contains the catalytic acid and catalytic base. A long V-shaped groove, partially enclosed at one end, forms a single extended substrate-binding surface across the face of the propeller. |
| 214757 | Glyco_32 | 2.18e-36 | 47 | 593 | 1 | 435 | Glycosyl hydrolases family 32. |
| 400517 | Glyco_hydro_32C | 2.18e-27 | 439 | 631 | 1 | 162 | Glycosyl hydrolases family 32 C terminal. This domain corresponds to the C terminal domain of glycosyl hydrolase family 32. It forms a beta sandwich module. |
| 395193 | Glyco_hydro_32N | 4.76e-20 | 103 | 398 | 58 | 298 | Glycosyl hydrolases family 32 N-terminal domain. This domain corresponds to the N-terminal domain of glycosyl hydrolase family 32 which forms a five bladed beta propeller structure. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| 0.0 | 1 | 642 | 1 | 663 | |
| 1.23e-315 | 10 | 641 | 10 | 694 | |
| 3.10e-315 | 10 | 641 | 10 | 681 | |
| 3.10e-315 | 10 | 641 | 10 | 681 | |
| 3.10e-315 | 10 | 641 | 10 | 681 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 1.12e-275 | 22 | 641 | 6 | 604 | Aspergillus kawachii beta-fructofuranosidase complexed with glycerol [Aspergillus luchuensis IFO 4308],5XH9_A Aspergillus kawachii beta-fructofuranosidase [Aspergillus luchuensis IFO 4308],5XHA_A Aspergillus kawachii beta-fructofuranosidase complexed with fructose [Aspergillus luchuensis IFO 4308] |
|
| 9.90e-245 | 26 | 640 | 10 | 634 | Crystal structure of fructosyltransferase (wild-type) from A. japonicus [Aspergillus japonicus],3LFI_A Crystal structure of fructosyltransferase (wild-type) from A. japonicus in complex with glucose [Aspergillus japonicus],3LFI_B Crystal structure of fructosyltransferase (wild-type) from A. japonicus in complex with glucose [Aspergillus japonicus] |
|
| 1.62e-243 | 26 | 640 | 10 | 634 | Crystal Structure of A. japonicus CB05 [Aspergillus japonicus],3LDR_A Crystal structure of fructosyltransferase (D191A) from A. japonicus in complex with 1-Kestose [Aspergillus japonicus],3LEM_A Crystal structure of fructosyltransferase (D191A) from A. japonicus in complex with Nystose [Aspergillus japonicus],3LIG_A Crystal structure of fructosyltransferase (D191A) from A. japonicus [Aspergillus japonicus],3LIH_A Crystal structure of fructosyltransferase (D191A) from A. japonicus in complex with raffinose [Aspergillus japonicus] |
|
| 1.30e-78 | 22 | 640 | 45 | 641 | Chain A, Beta-fructofuranosidase [Phaffia rhodozyma],5ANN_B Chain B, Beta-fructofuranosidase [Phaffia rhodozyma] |
|
| 1.30e-78 | 22 | 640 | 45 | 641 | Chain A, Beta-fructofuranosidase [Phaffia rhodozyma],6S82_B Chain B, Beta-fructofuranosidase [Phaffia rhodozyma] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 1.49e-06 | 17 | 213 | 25 | 210 | Invertase OS=Kluyveromyces lactis (strain ATCC 8585 / CBS 2359 / DSM 70799 / NBRC 1267 / NRRL Y-1140 / WM37) OX=284590 GN=INV1 PE=3 SV=1 |
|
| 2.93e-06 | 41 | 395 | 22 | 318 | Raffinose invertase OS=Escherichia coli OX=562 GN=rafD PE=3 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as SP
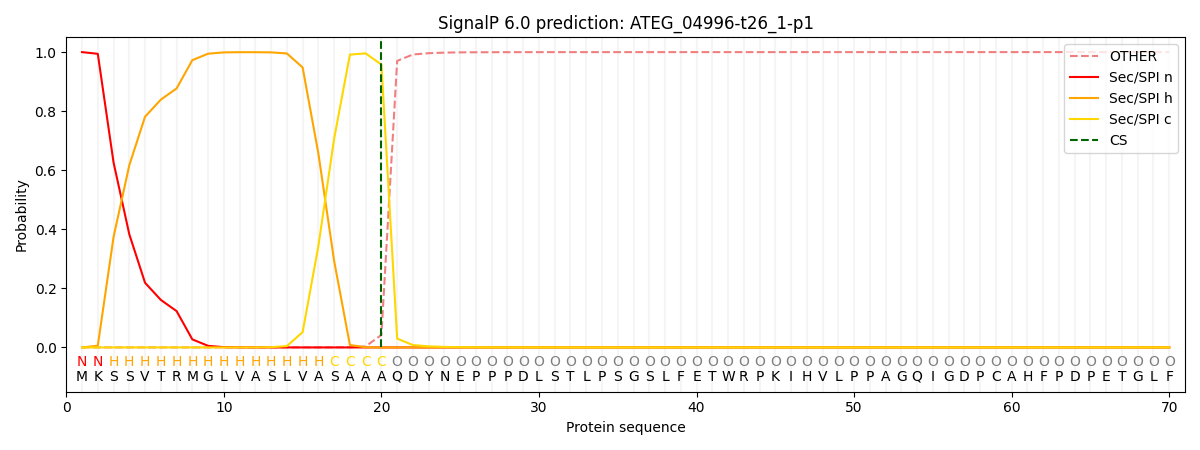
| Other | SP_Sec_SPI | CS Position |
|---|---|---|
| 0.000776 | 0.999191 | CS pos: 20-21. Pr: 0.9576 |
