You are browsing environment: FUNGIDB
CAZyme Information: ATEG_02541-t26_1-p1
You are here: Home > Sequence: ATEG_02541-t26_1-p1
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Aspergillus terreus | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Ascomycota; Eurotiomycetes; ; Aspergillaceae; Aspergillus; Aspergillus terreus | |||||||||||
| CAZyme ID | ATEG_02541-t26_1-p1 | |||||||||||
| CAZy Family | PL4 | |||||||||||
| CAZyme Description | conserved hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | ||||||||||||
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| 404688 | Glyco_transf_41 | 1.80e-104 | 1089 | 1552 | 28 | 543 | Glycosyl transferase family 41. This family of glycosyltransferases includes O-linked beta-N-acetylglucosamine (O-GlcNAc) transferase, an enzyme which catalyzes the addition of O-GlcNAc to serine and threonine residues. In addition to its function as an O-GlcNAc transferase, human OGT also appears to proteolytically cleave the epigenetic cell-cycle regulator HCF-1. |
| 226428 | Spy | 3.82e-65 | 905 | 1562 | 56 | 617 | Predicted O-linked N-acetylglucosamine transferase, SPINDLY family [Posttranslational modification, protein turnover, chaperones]. |
| 276809 | TPR | 6.25e-14 | 923 | 1001 | 1 | 79 | Tetratricopeptide repeat. The Tetratricopeptide repeat (TPR) typically contains 34 amino acids and is found in a variety of organisms including bacteria, cyanobacteria, yeast, fungi, plants, and humans. It is present in a variety of proteins including those involved in chaperone, cell-cycle, transcription, and protein transport complexes. The number of TPR motifs varies among proteins. Those containing 5-6 tandem repeats generate a right-handed helical structure with an amphipathic channel that is thought to accommodate an alpha-helix of a target protein. It has been proposed that TPR proteins preferentially interact with WD-40 repeat proteins, but in many instances several TPR-proteins seem to aggregate to multi-protein complexes. |
| 276809 | TPR | 7.74e-14 | 908 | 985 | 20 | 97 | Tetratricopeptide repeat. The Tetratricopeptide repeat (TPR) typically contains 34 amino acids and is found in a variety of organisms including bacteria, cyanobacteria, yeast, fungi, plants, and humans. It is present in a variety of proteins including those involved in chaperone, cell-cycle, transcription, and protein transport complexes. The number of TPR motifs varies among proteins. Those containing 5-6 tandem repeats generate a right-handed helical structure with an amphipathic channel that is thought to accommodate an alpha-helix of a target protein. It has been proposed that TPR proteins preferentially interact with WD-40 repeat proteins, but in many instances several TPR-proteins seem to aggregate to multi-protein complexes. |
| 276809 | TPR | 3.34e-11 | 539 | 624 | 8 | 93 | Tetratricopeptide repeat. The Tetratricopeptide repeat (TPR) typically contains 34 amino acids and is found in a variety of organisms including bacteria, cyanobacteria, yeast, fungi, plants, and humans. It is present in a variety of proteins including those involved in chaperone, cell-cycle, transcription, and protein transport complexes. The number of TPR motifs varies among proteins. Those containing 5-6 tandem repeats generate a right-handed helical structure with an amphipathic channel that is thought to accommodate an alpha-helix of a target protein. It has been proposed that TPR proteins preferentially interact with WD-40 repeat proteins, but in many instances several TPR-proteins seem to aggregate to multi-protein complexes. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| 0.0 | 1 | 1573 | 1 | 1668 | |
| 0.0 | 1 | 1573 | 1 | 1668 | |
| 0.0 | 1 | 1573 | 1 | 1668 | |
| 0.0 | 1 | 1573 | 1 | 1668 | |
| 0.0 | 1 | 1573 | 1 | 1670 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 4.12e-76 | 1089 | 1570 | 183 | 705 | O-GlcNAc transferase from Drososphila melanogaster [Drosophila melanogaster],5A01_B O-GlcNAc transferase from Drososphila melanogaster [Drosophila melanogaster],5A01_C O-GlcNAc transferase from Drososphila melanogaster [Drosophila melanogaster] |
|
| 2.55e-70 | 1089 | 1570 | 179 | 710 | The human O-GlcNAc transferase in complex with a thiol-linked bisubstrate inhibitor [Homo sapiens] |
|
| 2.60e-70 | 1089 | 1570 | 180 | 711 | The human O-GlcNAc transferase in complex with a bisubstrate inhibitor [Homo sapiens] |
|
| 2.86e-70 | 1089 | 1570 | 185 | 716 | Chain A, UDP-N-acetylglucosamine--peptide N-acetylglucosaminyltransferase 110 kDa subunit [Homo sapiens],3PE3_B Chain B, UDP-N-acetylglucosamine--peptide N-acetylglucosaminyltransferase 110 kDa subunit [Homo sapiens],3PE3_C Chain C, UDP-N-acetylglucosamine--peptide N-acetylglucosaminyltransferase 110 kDa subunit [Homo sapiens],3PE3_D Chain D, UDP-N-acetylglucosamine--peptide N-acetylglucosaminyltransferase 110 kDa subunit [Homo sapiens],3PE4_A Structure of human O-GlcNAc transferase and its complex with a peptide substrate [Homo sapiens],3PE4_C Structure of human O-GlcNAc transferase and its complex with a peptide substrate [Homo sapiens],3TAX_A A Neutral Diphosphate Mimic Crosslinks the Active Site of Human O-GlcNAc Transferase [Homo sapiens],3TAX_C A Neutral Diphosphate Mimic Crosslinks the Active Site of Human O-GlcNAc Transferase [Homo sapiens],4AY5_A Human O-GlcNAc transferase (OGT) in complex with UDP and glycopeptide [Homo sapiens],4AY5_B Human O-GlcNAc transferase (OGT) in complex with UDP and glycopeptide [Homo sapiens],4AY5_C Human O-GlcNAc transferase (OGT) in complex with UDP and glycopeptide [Homo sapiens],4AY5_D Human O-GlcNAc transferase (OGT) in complex with UDP and glycopeptide [Homo sapiens],4AY6_A Human O-GlcNAc transferase (OGT) in complex with UDP-5SGlcNAc and substrate peptide [Homo sapiens],4AY6_B Human O-GlcNAc transferase (OGT) in complex with UDP-5SGlcNAc and substrate peptide [Homo sapiens],4AY6_C Human O-GlcNAc transferase (OGT) in complex with UDP-5SGlcNAc and substrate peptide [Homo sapiens],4AY6_D Human O-GlcNAc transferase (OGT) in complex with UDP-5SGlcNAc and substrate peptide [Homo sapiens],4CDR_A Human O-GlcNAc transferase in complex with a bisubstrate inhibitor, Goblin1 [Homo sapiens],4CDR_B Human O-GlcNAc transferase in complex with a bisubstrate inhibitor, Goblin1 [Homo sapiens],4CDR_C Human O-GlcNAc transferase in complex with a bisubstrate inhibitor, Goblin1 [Homo sapiens],4CDR_D Human O-GlcNAc transferase in complex with a bisubstrate inhibitor, Goblin1 [Homo sapiens],4GYW_A Crystal structure of human O-GlcNAc Transferase in complex with UDP and a glycopeptide [Homo sapiens],4GYW_C Crystal structure of human O-GlcNAc Transferase in complex with UDP and a glycopeptide [Homo sapiens],4GYY_A Crystal structure of human O-GlcNAc Transferase with UDP-5SGlcNAc and a peptide substrate [Homo sapiens],4GYY_C Crystal structure of human O-GlcNAc Transferase with UDP-5SGlcNAc and a peptide substrate [Homo sapiens],4GZ3_A Crystal structure of human O-GlcNAc Transferase with UDP and a thioglycopeptide [Homo sapiens],4GZ3_C Crystal structure of human O-GlcNAc Transferase with UDP and a thioglycopeptide [Homo sapiens],4GZ5_A Crystal structure of human O-GlcNAc Transferase with UDP-GlcNAc [Homo sapiens],4GZ5_B Crystal structure of human O-GlcNAc Transferase with UDP-GlcNAc [Homo sapiens],4GZ5_C Crystal structure of human O-GlcNAc Transferase with UDP-GlcNAc [Homo sapiens],4GZ5_D Crystal structure of human O-GlcNAc Transferase with UDP-GlcNAc [Homo sapiens],4GZ6_A Crystal structure of human O-GlcNAc Transferase with UDP-5SGlcNAc [Homo sapiens],4GZ6_B Crystal structure of human O-GlcNAc Transferase with UDP-5SGlcNAc [Homo sapiens],4GZ6_C Crystal structure of human O-GlcNAc Transferase with UDP-5SGlcNAc [Homo sapiens],4GZ6_D Crystal structure of human O-GlcNAc Transferase with UDP-5SGlcNAc [Homo sapiens],4N39_A Crystal structure of human O-GlcNAc transferase bound to a peptide from HCF-1 pro-repeat 2 (11-26) [Homo sapiens],4N3A_A Crystal Structure of human O-GlcNAc transferase bound to a peptide from HCF-1 pro-repeat 2 (1-26)E10A [Homo sapiens],4N3B_A Crystal Structure of human O-GlcNAc Transferase bound to a peptide from HCF-1 pro-repeat2(1-26)E10Q and UDP-5SGlcNAc [Homo sapiens],4N3C_A Crystal Structure of human O-GlcNAc Transferase bound to a peptide from HCF-1 pro-repeat2(1-26) and UDP-GlcNAc [Homo sapiens],4XI9_A Human OGT in complex with UDP-5S-GlcNAc and substrate peptide (RBL2) [Homo sapiens],4XI9_B Human OGT in complex with UDP-5S-GlcNAc and substrate peptide (RBL2) [Homo sapiens],4XI9_C Human OGT in complex with UDP-5S-GlcNAc and substrate peptide (RBL2) [Homo sapiens],4XI9_D Human OGT in complex with UDP-5S-GlcNAc and substrate peptide (RBL2) [Homo sapiens],4XIF_A Human OGT in complex with UDP-5S-GlcNAc and substrate peptide (keratin-7) [Homo sapiens],4XIF_B Human OGT in complex with UDP-5S-GlcNAc and substrate peptide (keratin-7) [Homo sapiens],4XIF_C Human OGT in complex with UDP-5S-GlcNAc and substrate peptide (keratin-7) [Homo sapiens],4XIF_D Human OGT in complex with UDP-5S-GlcNAc and substrate peptide (keratin-7) [Homo sapiens],5BNW_A The active site of O-GlcNAc transferase imposes constraints on substrate sequence [Homo sapiens],5C1D_A Human OGT in complex with UDP-5S-GlcNAc and substrate peptide (RB2L) [Homo sapiens],5VIE_A Chain A, UDP-N-acetylglucosamine--peptide N-acetylglucosaminyltransferase 110 kDa subunit [Homo sapiens],5VIE_C Chain C, UDP-N-acetylglucosamine--peptide N-acetylglucosaminyltransferase 110 kDa subunit [Homo sapiens],5VIF_A Electrophilic probes for deciphering substrate recognition by O-GlcNAc transferase [Homo sapiens],6E37_A O-GlcNAc Transferase in complex with covalent inhibitor [Homo sapiens],6MA1_A Crystal structure of human O-GlcNAc transferase bound to a peptide from HCF-1 pro-repeat 2 (11-26) and inhibitor 4a [Homo sapiens],6MA2_A Crystal structure of human O-GlcNAc transferase bound to a peptide from HCF-1 pro-repeat 2 (11-26) and inhibitor ent-1a [Homo sapiens],6MA3_A Crystal structure of human O-GlcNAc transferase bound to a peptide from HCF-1 pro-repeat 2 (11-26) and inhibitor 2a [Homo sapiens],6MA4_A Crystal structure of human O-GlcNAc transferase bound to a peptide from HCF-1 pro-repeat 2 (11-26) and inhibitor 3a [Homo sapiens],6MA5_A Crystal structure of human O-GlcNAc transferase bound to a peptide from HCF-1 pro-repeat 2 (11-26) and inhibitor 1a [Homo sapiens] |
|
| 3.08e-70 | 1089 | 1570 | 184 | 715 | Chain AAA, UDP-N-acetylglucosamine--peptide N-acetylglucosaminyltransferase 110 kDa subunit [Homo sapiens] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 1.19e-85 | 1089 | 1568 | 535 | 960 | Probable UDP-N-acetylglucosamine--peptide N-acetylglucosaminyltransferase SEC OS=Arabidopsis thaliana OX=3702 GN=SEC PE=1 SV=1 |
|
| 4.30e-69 | 1089 | 1570 | 503 | 1034 | UDP-N-acetylglucosamine--peptide N-acetylglucosaminyltransferase 110 kDa subunit OS=Mus musculus OX=10090 GN=Ogt PE=1 SV=2 |
|
| 1.40e-68 | 1142 | 1566 | 663 | 1135 | UDP-N-acetylglucosamine--peptide N-acetylglucosaminyltransferase OS=Caenorhabditis elegans OX=6239 GN=ogt-1 PE=1 SV=2 |
|
| 1.02e-67 | 1089 | 1570 | 503 | 1034 | UDP-N-acetylglucosamine--peptide N-acetylglucosaminyltransferase 110 kDa subunit OS=Oryctolagus cuniculus OX=9986 GN=OGT PE=1 SV=2 |
|
| 1.35e-67 | 1089 | 1570 | 503 | 1034 | UDP-N-acetylglucosamine--peptide N-acetylglucosaminyltransferase 110 kDa subunit OS=Homo sapiens OX=9606 GN=OGT PE=1 SV=3 |
SignalP and Lipop Annotations help
This protein is predicted as OTHER
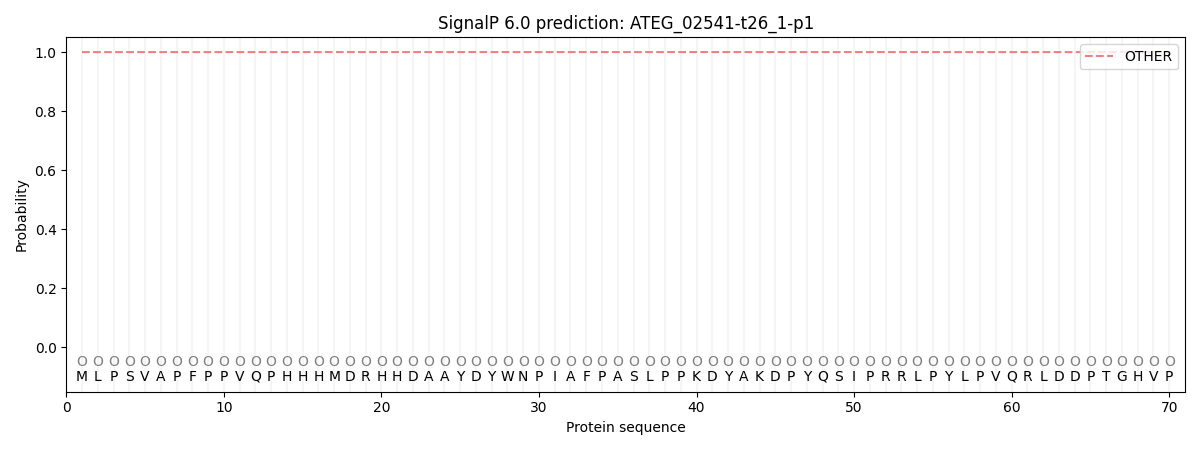
| Other | SP_Sec_SPI | CS Position |
|---|---|---|
| 1.000058 | 0.000000 |
