You are browsing environment: FUNGIDB
CAZyme Information: ASPGLDRAFT_52685-t33_1-p1
You are here: Home > Sequence: ASPGLDRAFT_52685-t33_1-p1
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Aspergillus glaucus | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Ascomycota; Eurotiomycetes; ; Aspergillaceae; Aspergillus; Aspergillus glaucus | |||||||||||
| CAZyme ID | ASPGLDRAFT_52685-t33_1-p1 | |||||||||||
| CAZy Family | GT1 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | ||||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| AA1 | 35 | 565 | 9e-78 | 0.9776536312849162 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| 259965 | CuRO_3_Abr2_like | 2.06e-72 | 408 | 572 | 1 | 164 | The third cupredoxin domain of a group of fungal Laccases similar to Abr2 from Aspergillus fumigatus. Abr2 is involved in conidial pigment biosynthesis in Aspergillus fumigatus. Laccase is a blue multi-copper enzyme that catalyzes the oxidation of a variety aromatic - notably phenolic and inorganic substances coupled to the reduction of molecular oxygen to water. Laccase has been implicated in a wide spectrum of biological activities and, in particular, plays a key role in morphogenesis, development and lignin metabolism in fungi and plants. Like other related multicopper oxidases (MCOs), laccase is composed of three cupredoxin domains that include one mononuclear and one trinuclear copper center. The copper ions are bound in several sites: Type 1, Type 2, and/or Type 3. The ensemble of types 2 and 3 copper is called a trinuclear cluster. MCOs oxidize their substrate by accepting electrons at a mononuclear copper center and transferring them to the active site trinuclear copper center. The cupredoxin domain 3 of 3-domain MCOs contains the Type 1 (T1) copper binding site and part the trinuclear copper binding site, which is located at the interface of domains 1 and 3. |
| 259944 | CuRO_2_Abr2_like | 1.77e-63 | 168 | 363 | 3 | 138 | The second cupredoxin domain of a group of fungal Laccases similar to Abr2 from Aspergillus fumigatus. Abr2 is involved in conidial pigment biosynthesis in Aspergillus fumigatus. Laccase is a blue multi-copper enzyme that catalyzes the oxidation of a variety aromatic - notably phenolic and inorganic substances coupled to the reduction of molecular oxygen to water. Laccase has been implicated in a wide spectrum of biological activities and, in particular, plays a key role in morphogenesis, development and lignin metabolism in fungi and plants. Like other related multicopper oxidases (MCOs), laccase is composed of three cupredoxin domains that include one mononuclear and one trinuclear copper center. The copper ions are bound in several sites: Type 1, Type 2, and/or Type 3. The ensemble of types 2 and 3 copper is called a trinuclear cluster. MCOs oxidize their substrate by accepting electrons at a mononuclear copper center and transferring them to the active site trinuclear copper center. The cupredoxin domain 2 of 3-domain MCOs has lost the ability to bind copper. |
| 274555 | ascorbase | 1.89e-58 | 20 | 584 | 3 | 536 | L-ascorbate oxidase, plant type. Members of this protein family are the copper-containing enzyme L-ascorbate oxidase (EC 1.10.3.3), also called ascorbase. This family is found in flowering plants, and shows greater sequence similarity to a family of laccases (EC 1.10.3.2) from plants than to other known ascorbate oxidases. |
| 259919 | CuRO_1_Abr2_like | 4.57e-55 | 23 | 136 | 1 | 117 | The first cupredoxin domain of a group of fungal Laccases similar to Abr2 from Aspergillus fumigatus. Abr2 is involved in conidial pigment biosynthesis in Aspergillus fumigatus. Laccase is a blue multi-copper enzyme that catalyzes the oxidation of a variety aromatic - notably phenolic and inorganic substances coupled to the reduction of molecular oxygen to water. Laccase has been implicated in a wide spectrum of biological activities and, in particular, plays a key role in morphogenesis, development and lignin metabolism in fungi and plants. Like other related multicopper oxidases (MCOs), laccase is composed of three cupredoxin domains that include one mononuclear and one trinuclear copper center. The copper ions are bound in several sites: Type 1, Type 2, and/or Type 3. The ensemble of types 2 and 3 copper is called a trinuclear cluster. MCOs oxidize their substrate by accepting electrons at a mononuclear copper center and transferring them to the active site trinuclear copper center. The cupredoxin domain 1 of 3-domain MCOs contains part the trinuclear copper binding site, which is located at the interface of domains 1 and 3. |
| 177843 | PLN02191 | 1.89e-54 | 14 | 584 | 19 | 559 | L-ascorbate oxidase |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| 0.0 | 1 | 583 | 1 | 583 | |
| 4.02e-291 | 11 | 584 | 15 | 586 | |
| 2.29e-239 | 48 | 582 | 3 | 539 | |
| 4.44e-207 | 16 | 583 | 21 | 591 | |
| 4.44e-207 | 16 | 583 | 21 | 591 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 9.39e-46 | 20 | 584 | 5 | 536 | Refined Crystal Structure Of Ascorbate Oxidase At 1.9 Angstroms Resolution [Cucurbita pepo var. melopepo],1AOZ_B Refined Crystal Structure Of Ascorbate Oxidase At 1.9 Angstroms Resolution [Cucurbita pepo var. melopepo],1ASO_A X-Ray Structures And Mechanistic Implications Of Three Functional Derivatives Of Ascorbate Oxidase From Zucchini: Reduced-, Peroxide-, And Azide-Forms [Cucurbita pepo var. melopepo],1ASO_B X-Ray Structures And Mechanistic Implications Of Three Functional Derivatives Of Ascorbate Oxidase From Zucchini: Reduced-, Peroxide-, And Azide-Forms [Cucurbita pepo var. melopepo],1ASP_A X-ray Structures And Mechanistic Implications Of Three Functional Derivatives Of Ascorbate Oxidase From Zucchini: Reduced-, Peroxide-, And Azide-forms [Cucurbita pepo var. melopepo],1ASP_B X-ray Structures And Mechanistic Implications Of Three Functional Derivatives Of Ascorbate Oxidase From Zucchini: Reduced-, Peroxide-, And Azide-forms [Cucurbita pepo var. melopepo],1ASQ_A X-Ray Structures And Mechanistic Implications Of Three Functional Derivatives Of Ascorbate Oxidase From Zucchini: Reduced-, Peroxide-, And Azide-Forms [Cucurbita pepo var. melopepo],1ASQ_B X-Ray Structures And Mechanistic Implications Of Three Functional Derivatives Of Ascorbate Oxidase From Zucchini: Reduced-, Peroxide-, And Azide-Forms [Cucurbita pepo var. melopepo] |
|
| 9.09e-45 | 24 | 583 | 7 | 488 | Crystal Structure of Blue Laccase from Trametes trogii complexed with p-methylbenzoate [Coriolopsis trogii],2HRH_A Crystal Structure of Blue Laccase from Trametes trogii [Coriolopsis trogii] |
|
| 6.17e-44 | 23 | 583 | 8 | 488 | T2-depleted laccase from Coriolopsis caperata soaked with CuCl [Coriolopsis caperata],4JHV_A T2-depleted laccase from Coriolopsis caperata [Coriolopsis caperata] |
|
| 5.72e-43 | 24 | 583 | 7 | 488 | Crystal Structure Of Laccase From Basidiomycete Pm1 (cect 2971) [basidiomycete PM1],5ANH_B Crystal Structure Of Laccase From Basidiomycete Pm1 (cect 2971) [basidiomycete PM1],5ANH_C Crystal Structure Of Laccase From Basidiomycete Pm1 (cect 2971) [basidiomycete PM1] |
|
| 5.72e-43 | 24 | 583 | 7 | 488 | Coriolopsis gallica Laccase T2 Copper Depleted at pH 4.5 [Coriolopsis gallica],4A2E_A Crystal Structure of a Coriolopsis gallica Laccase at 1.7 A Resolution pH 5.5 [Coriolopsis gallica],4A2G_A Coriolopsis gallica laccase collected at 8.98 keV [Coriolopsis gallica],4A2H_A Crystal Structure of Laccase from Coriolopsis gallica pH 7.0 [Coriolopsis gallica] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 5.73e-289 | 10 | 579 | 11 | 579 | Laccase abr2 OS=Neosartorya fumigata (strain ATCC MYA-4609 / Af293 / CBS 101355 / FGSC A1100) OX=330879 GN=abr2 PE=1 SV=1 |
|
| 5.78e-179 | 12 | 583 | 18 | 600 | Multicopper oxidase GIP1 OS=Gibberella zeae (strain ATCC MYA-4620 / CBS 123657 / FGSC 9075 / NRRL 31084 / PH-1) OX=229533 GN=GIP1 PE=1 SV=1 |
|
| 1.54e-175 | 17 | 583 | 18 | 597 | Multicopper oxidase VdtB OS=Byssochlamys spectabilis OX=264951 GN=VdtB PE=1 SV=1 |
|
| 7.40e-171 | 5 | 583 | 6 | 605 | Laccase ustL OS=Ustilaginoidea virens OX=1159556 GN=ustL PE=1 SV=1 |
|
| 2.25e-165 | 13 | 583 | 18 | 592 | Multicopper oxidase MCE OS=Talaromyces pinophilus OX=128442 GN=MCE PE=1 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as SP
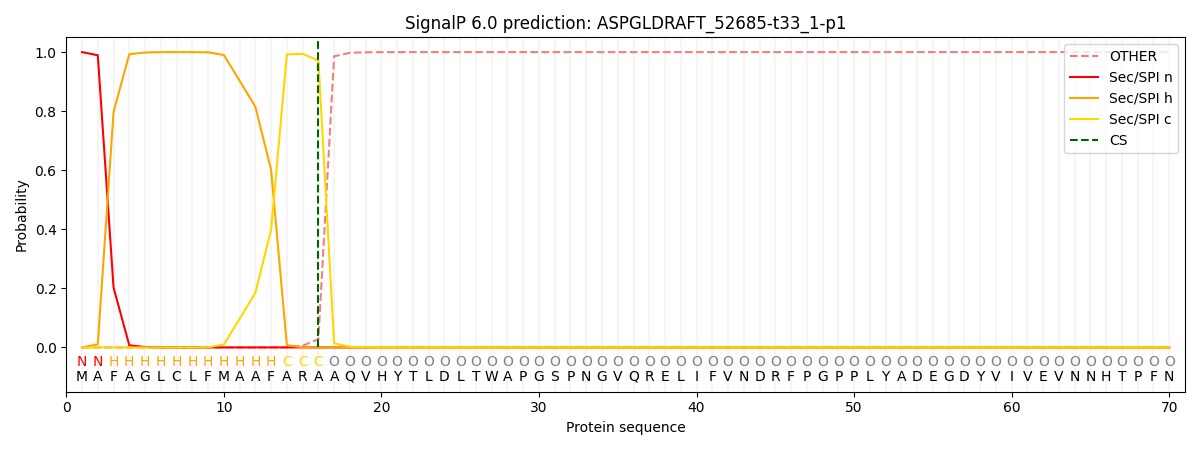
| Other | SP_Sec_SPI | CS Position |
|---|---|---|
| 0.000241 | 0.999712 | CS pos: 16-17. Pr: 0.9717 |
