You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000003363_02997
You are here: Home > Sequence: MGYG000003363_02997
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
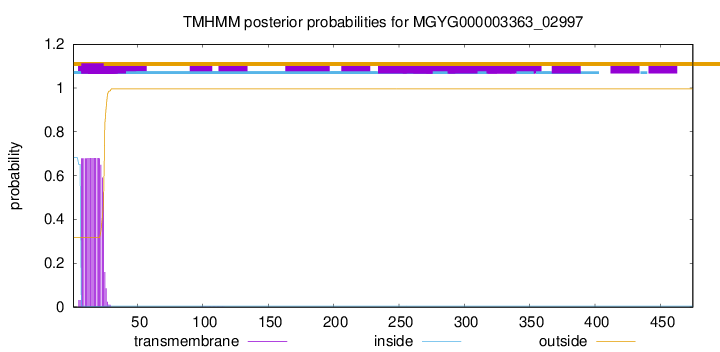
TMHMM annotations
Basic Information help
| Species | Bacteroides sp900766195 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Bacteroidota; Bacteroidia; Bacteroidales; Bacteroidaceae; Bacteroides; Bacteroides sp900766195 | |||||||||||
| CAZyme ID | MGYG000003363_02997 | |||||||||||
| CAZy Family | GH27 | |||||||||||
| CAZyme Description | Isomalto-dextranase | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 5379; End: 6806 Strand: - | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH27 | 194 | 453 | 1e-33 | 0.9388646288209607 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| pfam17801 | Melibiase_C | 1.34e-15 | 400 | 473 | 1 | 74 | Alpha galactosidase C-terminal beta sandwich domain. This domain is found at the C-terminus of alpha galactosidase enzymes. |
| PLN02808 | PLN02808 | 7.52e-10 | 342 | 474 | 249 | 384 | alpha-galactosidase |
| PLN02229 | PLN02229 | 8.71e-09 | 257 | 474 | 210 | 418 | alpha-galactosidase |
| cd14792 | GH27 | 9.64e-09 | 74 | 387 | 14 | 271 | glycosyl hydrolase family 27 (GH27). GH27 enzymes occur in eukaryotes, prokaryotes, and archaea with a wide range of hydrolytic activities, including alpha-glucosidase (glucoamylase and sucrase-isomaltase), alpha-N-acetylgalactosaminidase, and 3-alpha-isomalto-dextranase. All GH27 enzymes cleave a terminal carbohydrate moiety from a substrate that varies considerably in size, depending on the enzyme, and may be either a starch or a glycoprotein. GH27 members are retaining enzymes that cleave their substrates via an acid/base-catalyzed, double-displacement mechanism involving a covalent glycosyl-enzyme intermediate. Two aspartic acid residues have been identified as the catalytic nucleophile and the acid/base, respectively. |
| PLN02692 | PLN02692 | 3.02e-05 | 87 | 473 | 83 | 408 | alpha-galactosidase |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| QUU07210.1 | 2.27e-226 | 44 | 475 | 52 | 492 |
| QUT44530.1 | 6.49e-226 | 44 | 475 | 52 | 492 |
| QUT79686.1 | 6.49e-226 | 44 | 475 | 52 | 492 |
| QRM98947.1 | 6.49e-226 | 44 | 475 | 52 | 492 |
| QNT67171.1 | 1.71e-214 | 3 | 473 | 2 | 473 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 5AWO_A | 4.91e-89 | 50 | 474 | 37 | 466 | Arthrobacterglobiformis T6 isomalto-dextranse [Arthrobacter globiformis],5AWP_A Arthrobacter globiformis T6 isomalto-dextranase complexed with isomaltose [Arthrobacter globiformis],5AWQ_A Arthrobacter globiformis T6 isomalto-dextranse complexed with panose [Arthrobacter globiformis] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| Q44052 | 5.25e-89 | 50 | 474 | 63 | 492 | Isomalto-dextranase OS=Arthrobacter globiformis OX=1665 GN=imd PE=1 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as LIPO
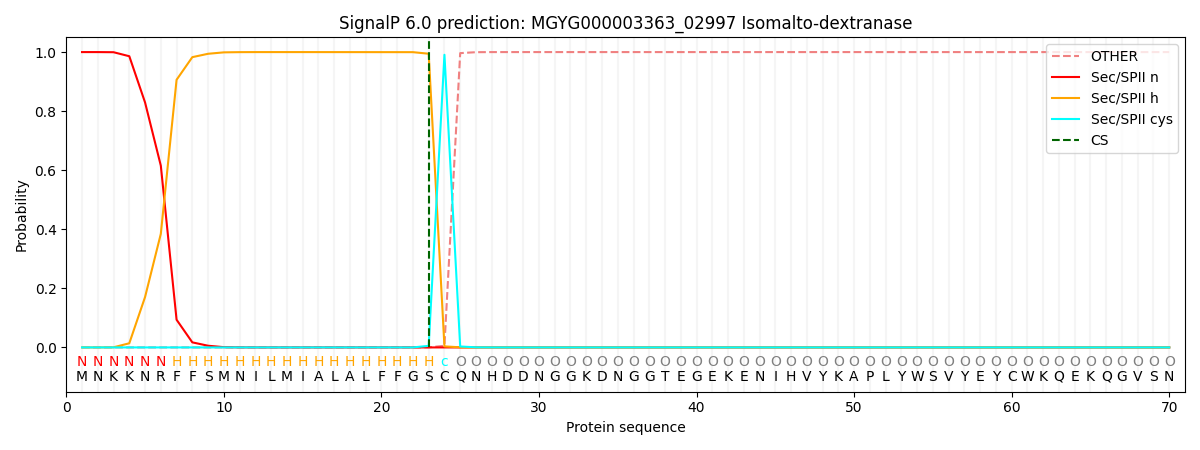
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.000000 | 0.000012 | 1.000040 | 0.000000 | 0.000000 | 0.000000 |