You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000002963_03807
You are here: Home > Sequence: MGYG000002963_03807
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
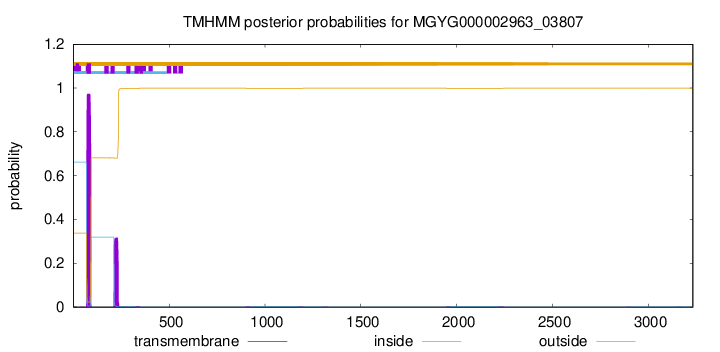
TMHMM annotations
Basic Information help
| Species | Anaerosporobacter mobilis | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Firmicutes_A; Clostridia; Lachnospirales; Lachnospiraceae; Anaerosporobacter; Anaerosporobacter mobilis | |||||||||||
| CAZyme ID | MGYG000002963_03807 | |||||||||||
| CAZy Family | GT2 | |||||||||||
| CAZyme Description | Tyrocidine synthase 2 | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 13138; End: 22839 Strand: - | |||||||||||
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| cd17655 | A_NRPS_Bac | 0.0 | 1123 | 1618 | 1 | 489 | bacitracin synthetase and related proteins. This family of the adenylation (A) domain of nonribosomal peptide synthases (NRPS) includes bacitracin synthetases 1, 2, and 3 (BA1, also known as ATP-dependent cysteine adenylase or cysteine activase, BA2, also known as ATP-dependent lysine adenylase or lysine activase, and BA3, also known as ATP-dependent isoleucine adenylase or isoleucine activase) in Bacilli. Bacitracin is a mixture of related cyclic peptides used as a polypeptide antibiotic. This family also includes gramicidin synthetase 1 involved in synthesis of the cyclic peptide antibiotic gramicidin S via activation of phenylalanine. NRPSs are large multifunctional enzymes which synthesize many therapeutically useful peptides in bacteria and fungi via a template-directed, nucleic acid independent nonribosomal mechanism. These natural products include antibiotics, immunosuppressants, plant and animal toxins, and enzyme inhibitors. NRPS has a distinct modular structure in which each module is responsible for the recognition, activation, and in some cases, modification of a single amino acid residue of the final peptide product. The modules can be subdivided into domains that catalyze specific biochemical reactions. |
| cd17655 | A_NRPS_Bac | 0.0 | 2168 | 2670 | 1 | 490 | bacitracin synthetase and related proteins. This family of the adenylation (A) domain of nonribosomal peptide synthases (NRPS) includes bacitracin synthetases 1, 2, and 3 (BA1, also known as ATP-dependent cysteine adenylase or cysteine activase, BA2, also known as ATP-dependent lysine adenylase or lysine activase, and BA3, also known as ATP-dependent isoleucine adenylase or isoleucine activase) in Bacilli. Bacitracin is a mixture of related cyclic peptides used as a polypeptide antibiotic. This family also includes gramicidin synthetase 1 involved in synthesis of the cyclic peptide antibiotic gramicidin S via activation of phenylalanine. NRPSs are large multifunctional enzymes which synthesize many therapeutically useful peptides in bacteria and fungi via a template-directed, nucleic acid independent nonribosomal mechanism. These natural products include antibiotics, immunosuppressants, plant and animal toxins, and enzyme inhibitors. NRPS has a distinct modular structure in which each module is responsible for the recognition, activation, and in some cases, modification of a single amino acid residue of the final peptide product. The modules can be subdivided into domains that catalyze specific biochemical reactions. |
| PRK12467 | PRK12467 | 0.0 | 667 | 3232 | 42 | 2644 | peptide synthase; Provisional |
| PRK12316 | PRK12316 | 0.0 | 675 | 3232 | 1557 | 4100 | peptide synthase; Provisional |
| cd05930 | A_NRPS | 0.0 | 2178 | 2666 | 1 | 444 | The adenylation domain of nonribosomal peptide synthetases (NRPS). The adenylation (A) domain of NRPS recognizes a specific amino acid or hydroxy acid and activates it as an (amino) acyl adenylate by hydrolysis of ATP. The activated acyl moiety then forms a thioester bond to the enzyme-bound cofactor phosphopantetheine of a peptidyl carrier protein domain. NRPSs are large multifunctional enzymes which synthesize many therapeutically useful peptides in bacteria and fungi via a template-directed, nucleic acid independent nonribosomal mechanism. These natural products include antibiotics, immunosuppressants, plant and animal toxins, and enzyme inhibitors. NRPS has a distinct modular structure in which each module is responsible for the recognition, activation, and in some cases, modification of a single amino acid residue of the final peptide product. The modules can be subdivided into domains that catalyze specific biochemical reactions. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| QND46664.1 | 0.0 | 170 | 2781 | 74 | 2691 |
| BAY90071.1 | 3.83e-195 | 575 | 1762 | 2123 | 3353 |
| BAZ00088.1 | 5.17e-195 | 542 | 1762 | 2096 | 3362 |
| BAZ75991.1 | 5.17e-195 | 542 | 1762 | 2096 | 3362 |
| BAY30132.1 | 1.20e-194 | 546 | 1762 | 2099 | 3364 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 6MFZ_A | 0.0 | 1120 | 2757 | 206 | 1802 | Crystalstructure of dimodular LgrA in a condensation state [Brevibacillus parabrevis],6MFZ_B Crystal structure of dimodular LgrA in a condensation state [Brevibacillus parabrevis] |
| 6MFY_A | 3.17e-310 | 1120 | 2670 | 206 | 1716 | Crystalstructure of a 5-domain construct of LgrA in the substrate donation state [Brevibacillus parabrevis],6MG0_A Crystal structure of a 5-domain construct of LgrA in the thiolation state [Brevibacillus parabrevis],6MG0_B Crystal structure of a 5-domain construct of LgrA in the thiolation state [Brevibacillus parabrevis] |
| 6MFW_A | 5.01e-191 | 1120 | 2135 | 206 | 1195 | Crystalstructure of a 4-domain construct of LgrA in the substrate donation state [Brevibacillus parabrevis] |
| 6MFX_A | 2.31e-190 | 1120 | 2135 | 206 | 1195 | Crystalstructure of a 4-domain construct of a mutant of LgrA in the substrate donation state [Brevibacillus parabrevis] |
| 5U89_A | 2.69e-177 | 1099 | 2152 | 5 | 1073 | Crystalstructure of a cross-module fragment from the dimodular NRPS DhbF [Geobacillus sp. Y4.1MC1] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| P39847 | 0.0 | 667 | 3231 | 3 | 2555 | Plipastatin synthase subunit C OS=Bacillus subtilis (strain 168) OX=224308 GN=ppsC PE=1 SV=2 |
| Q04747 | 0.0 | 12 | 3230 | 473 | 3580 | Surfactin synthase subunit 2 OS=Bacillus subtilis (strain 168) OX=224308 GN=srfAB PE=1 SV=3 |
| P94459 | 0.0 | 33 | 3228 | 489 | 3600 | Plipastatin synthase subunit D OS=Bacillus subtilis (strain 168) OX=224308 GN=ppsD PE=1 SV=2 |
| Q70LM4 | 0.0 | 674 | 3211 | 7 | 2558 | Linear gramicidin synthase subunit D OS=Brevibacillus parabrevis OX=54914 GN=lgrD PE=1 SV=1 |
| P39846 | 0.0 | 667 | 3221 | 3 | 2550 | Plipastatin synthase subunit B OS=Bacillus subtilis (strain 168) OX=224308 GN=ppsB PE=1 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as OTHER
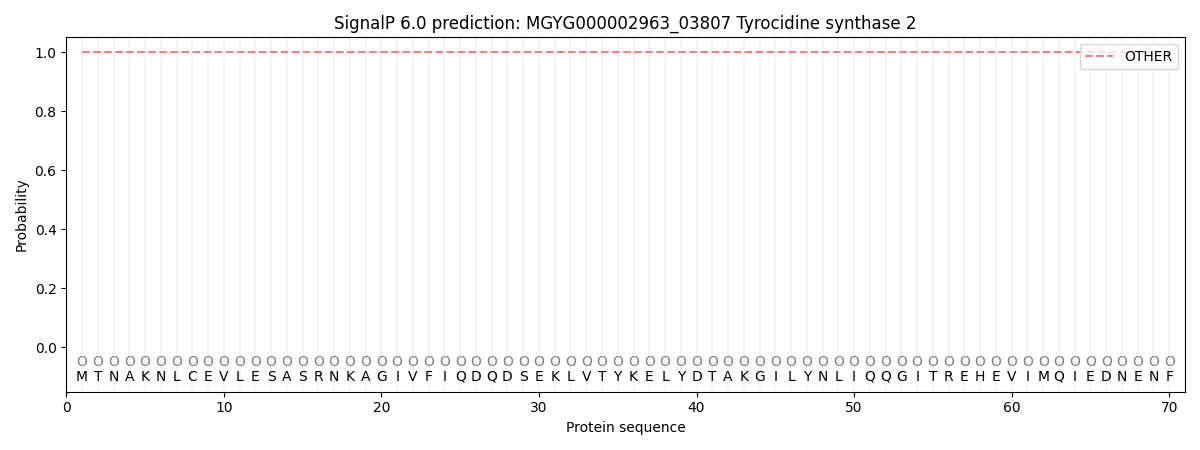
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 1.000086 | 0.000000 | 0.000000 | 0.000000 | 0.000000 | 0.000000 |