You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000002585_00566
You are here: Home > Sequence: MGYG000002585_00566
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Bacteroidota; Bacteroidia; Bacteroidales; Bacteroidaceae; Bacteroides; | |||||||||||
| CAZyme ID | MGYG000002585_00566 | |||||||||||
| CAZy Family | GH10 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 48337; End: 51063 Strand: + | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH43 | 399 | 693 | 4.7e-114 | 0.9928825622775801 |
| GH10 | 33 | 370 | 1.3e-102 | 0.9867986798679867 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| cd18617 | GH43_XynB-like | 2.04e-164 | 399 | 695 | 1 | 285 | Glycosyl hydrolase family 43, such as Bacteroides ovatus alpha-L-arabinofuranosidase (BoGH43, XynB). This glycosyl hydrolase family 43 (GH43) subgroup includes enzymes that have been characterized to have alpha-L-arabinofuranosidase (EC 3.2.1.55) and beta-1,4-xylosidase (beta-D-xylosidase;xylan 1,4-beta-xylosidase; EC 3.2.1.37) activities. Beta-1,4-xylosidases are part of an array of hemicellulases that are involved in the final breakdown of plant cell-wall whereby they degrade xylan. They hydrolyze beta-1,4 glycosidic bonds between two xylose units in short xylooligosaccharides. These are inverting enzymes (i.e. they invert the stereochemistry of the anomeric carbon atom of the substrate) that have an aspartate as the catalytic general base, a glutamate as the catalytic general acid and another aspartate that is responsible for pKa modulation and orienting the catalytic acid. Also included in this subfamily are Bacteroides ovatus alpha-L-arabinofuranosidases, BoGH43A and BoGH43B, both having a two-domain architecture, consisting of an N-terminal 5-bladed beta-propeller domain harboring the catalytic active site, and a C-terminal beta-sandwich domain. However, despite significant functional overlap between these two enzymes, BoGH43A and BoGH43B share just 41% sequence identity. The latter appears to be significantly less active on the same substrates, suggesting that these paralogs may play subtly different roles during the degradation of xyloglucans from different sources, or may function most optimally at different stages in the catabolism of xyloglucan oligosaccharides (XyGOs), for example before or after hydrolysis of certain side-chain moieties. It also includes Phanerochaete chrysosporium BKM-F-1767 Xyl, a bifunctional xylosidase/arabinofuranosidase. A common structural feature of GH43 enzymes is a 5-bladed beta-propeller domain that contains the catalytic acid and catalytic base. A long V-shaped groove, partially enclosed at one end, forms a single extended substrate-binding surface across the face of the propeller. |
| pfam00331 | Glyco_hydro_10 | 2.74e-111 | 33 | 364 | 1 | 303 | Glycosyl hydrolase family 10. |
| smart00633 | Glyco_10 | 9.88e-103 | 76 | 364 | 1 | 258 | Glycosyl hydrolase family 10. |
| COG3507 | XynB2 | 8.70e-99 | 399 | 906 | 23 | 534 | Beta-xylosidase [Carbohydrate transport and metabolism]. |
| cd09000 | GH43_SXA-like | 3.60e-92 | 399 | 693 | 1 | 290 | Glycosyl hydrolase family 43, such as Selenomonas ruminantium beta-D-xylosidase SXA. This glycosyl hydrolase family 43 (GH43) includes enzymes that have been characterized to mainly have beta-1,4-xylosidase (beta-D-xylosidase;xylan 1,4-beta-xylosidase; EC 3.2.1.37) activity, including Selenomonas ruminantium (Xsa;Sxa;SXA), Bifidobacterium adolescentis ATCC 15703 (XylC;XynB;BAD_0428) and Bacillus sp. KK-1 XylB. They are part of an array of hemicellulases that are involved in the final breakdown of plant cell-wall whereby they degrade xylan. They hydrolyze beta-1,4 glycosidic bonds between two xylose units in short xylooligosaccharides. These are inverting enzymes (i.e. they invert the stereochemistry of the anomeric carbon atom of the substrate) that have an aspartate as the catalytic general base, a glutamate as the catalytic general acid and another aspartate that is responsible for pKa modulation and orienting the catalytic acid. These enzymes possess an additional C-terminal beta-sandwich domain that restricts access for substrates to a portion of the active site to form a pocket. The active-site pockets comprise of two subsites, with binding capacity for two monosaccharide moieties and a single route of access for small molecules such as substrate. A common structural feature of GH43 enzymes is a 5-bladed beta-propeller domain that contains the catalytic acid and catalytic base. A long V-shaped groove, partially enclosed at one end, forms a single extended substrate-binding surface across the face of the propeller. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| ALJ61536.1 | 0.0 | 1 | 905 | 31 | 927 |
| QUT92894.1 | 0.0 | 19 | 905 | 3 | 882 |
| QDO69420.1 | 0.0 | 1 | 906 | 1 | 898 |
| EDV05059.1 | 0.0 | 1 | 906 | 1 | 909 |
| QCD38829.1 | 5.95e-207 | 375 | 902 | 30 | 547 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 7ERL_A | 6.13e-115 | 399 | 902 | 31 | 538 | ChainA, Beta-xylanase [Bacteroides intestinalis],7ERL_B Chain B, Beta-xylanase [Bacteroides intestinalis] |
| 4PMU_A | 6.38e-93 | 32 | 375 | 2 | 355 | Crystalstructure of a novel reducing-end xylose-releasing exo-oligoxylanase (XynA) belonging to GH10 family (space group P1211) [Xanthomonas citri pv. citri str. 306],4PMU_B Crystal structure of a novel reducing-end xylose-releasing exo-oligoxylanase (XynA) belonging to GH10 family (space group P1211) [Xanthomonas citri pv. citri str. 306],4PMU_C Crystal structure of a novel reducing-end xylose-releasing exo-oligoxylanase (XynA) belonging to GH10 family (space group P1211) [Xanthomonas citri pv. citri str. 306],4PMU_D Crystal structure of a novel reducing-end xylose-releasing exo-oligoxylanase (XynA) belonging to GH10 family (space group P1211) [Xanthomonas citri pv. citri str. 306],4PMU_E Crystal structure of a novel reducing-end xylose-releasing exo-oligoxylanase (XynA) belonging to GH10 family (space group P1211) [Xanthomonas citri pv. citri str. 306],4PMU_F Crystal structure of a novel reducing-end xylose-releasing exo-oligoxylanase (XynA) belonging to GH10 family (space group P1211) [Xanthomonas citri pv. citri str. 306] |
| 4PMV_A | 6.59e-93 | 32 | 375 | 3 | 356 | Crystalstructure of a novel reducing-end xylose-releasing exo-oligoxylanase (XynA) belonging to GH10 family (space group P43212) [Xanthomonas citri pv. citri str. 306],4PMV_B Crystal structure of a novel reducing-end xylose-releasing exo-oligoxylanase (XynA) belonging to GH10 family (space group P43212) [Xanthomonas citri pv. citri str. 306] |
| 6FHE_A | 5.38e-92 | 31 | 364 | 11 | 333 | Highlyactive enzymes by automated modular backbone assembly and sequence design [synthetic construct] |
| 1UQY_A | 2.59e-91 | 30 | 363 | 24 | 359 | XylanaseXyn10B mutant (E262S) from Cellvibrio mixtus in complex with xylopentaose [Cellvibrio mixtus],1UQZ_A Xylanase Xyn10B mutant (E262S) from Cellvibrio mixtus in complex with 4-O-methyl glucuronic acid [Cellvibrio mixtus],1UR1_A Xylanase Xyn10B mutant (E262S) from Cellvibrio mixtus in complex with arabinofuranose alpha-1,3 linked to xylobiose [Cellvibrio mixtus],1UR2_A Xylanase Xyn10B mutant (E262S) from Cellvibrio mixtus in complex with arabinofuranose alpha 1,3 linked to xylotriose [Cellvibrio mixtus] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| P49942 | 3.15e-171 | 1 | 370 | 1 | 369 | Endo-1,4-beta-xylanase A OS=Bacteroides ovatus OX=28116 GN=xylI PE=2 SV=1 |
| P48789 | 2.90e-114 | 30 | 370 | 23 | 365 | Endo-1,4-beta-xylanase A OS=Prevotella ruminicola OX=839 GN=xynA PE=3 SV=1 |
| P45982 | 1.25e-84 | 398 | 904 | 5 | 499 | Xylosidase/arabinosidase OS=Butyrivibrio fibrisolvens OX=831 GN=xylB PE=3 SV=1 |
| A7LXT8 | 1.14e-79 | 392 | 902 | 18 | 501 | Non-reducing end alpha-L-arabinofuranosidase BoGH43A OS=Bacteroides ovatus (strain ATCC 8483 / DSM 1896 / JCM 5824 / BCRC 10623 / CCUG 4943 / NCTC 11153) OX=411476 GN=BACOVA_02654 PE=1 SV=1 |
| A7LXU0 | 1.57e-74 | 399 | 908 | 29 | 514 | Non-reducing end alpha-L-arabinofuranosidase BoGH43B OS=Bacteroides ovatus (strain ATCC 8483 / DSM 1896 / JCM 5824 / BCRC 10623 / CCUG 4943 / NCTC 11153) OX=411476 GN=BACOVA_02656 PE=1 SV=2 |
SignalP and Lipop Annotations help
This protein is predicted as LIPO
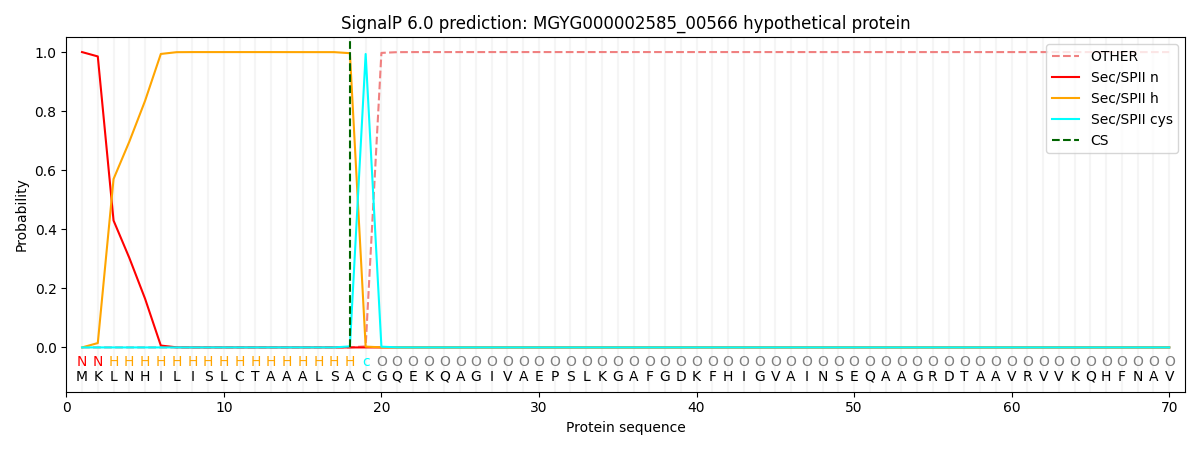
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.000000 | 0.000000 | 1.000093 | 0.000000 | 0.000000 | 0.000000 |
