You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000001554_00259
You are here: Home > Sequence: MGYG000001554_00259
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Corynebacterium phoceense | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Actinobacteriota; Actinomycetia; Mycobacteriales; Mycobacteriaceae; Corynebacterium; Corynebacterium phoceense | |||||||||||
| CAZyme ID | MGYG000001554_00259 | |||||||||||
| CAZy Family | GT2 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 268658; End: 273517 Strand: - | |||||||||||
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| COG3321 | PksD | 0.0 | 100 | 1056 | 1 | 931 | Acyl transferase domain in polyketide synthase (PKS) enzymes [Secondary metabolites biosynthesis, transport and catabolism]. |
| cd00833 | PKS | 0.0 | 104 | 523 | 2 | 421 | polyketide synthases (PKSs) polymerize simple fatty acids into a large variety of different products, called polyketides, by successive decarboxylating Claisen condensations. PKSs can be divided into 2 groups, modular type I PKSs consisting of one or more large multifunctional proteins and iterative type II PKSs, complexes of several monofunctional subunits. |
| smart00825 | PKS_KS | 8.80e-137 | 105 | 523 | 1 | 296 | Beta-ketoacyl synthase. The structure of beta-ketoacyl synthase is similar to that of the thiolase family and also chalcone synthase. The active site of beta-ketoacyl synthase is located between the N and C-terminal domains. |
| pfam00109 | ketoacyl-synt | 3.88e-78 | 105 | 350 | 1 | 249 | Beta-ketoacyl synthase, N-terminal domain. The structure of beta-ketoacyl synthase is similar to that of the thiolase family (pfam00108) and also chalcone synthase. The active site of beta-ketoacyl synthase is located between the N and C-terminal domains. The N-terminal domain contains most of the structures involved in dimer formation and also the active site cysteine. |
| TIGR02813 | omega_3_PfaA | 1.82e-75 | 105 | 920 | 9 | 871 | polyketide-type polyunsaturated fatty acid synthase PfaA. Members of the seed for this alignment are involved in omega-3 polyunsaturated fatty acid biosynthesis, such as the protein PfaA from the eicosapentaenoic acid biosynthesis operon in Photobacterium profundum strain SS9. PfaA is encoded together with PfaB, PfaC, and PfaD, and the functions of the individual polypeptides have not yet been described. More distant homologs of PfaA, also included with the reach of this model, appear to be involved in polyketide-like biosynthetic mechanisms of polyunsaturated fatty acid biosynthesis, an alternative to the more familiar iterated mechanism of chain extension and desaturation, and in most cases are encoded near genes for homologs of PfaB, PfaC, and/or PfaD. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| BAZ00088.1 | 3.74e-107 | 44 | 917 | 1148 | 2015 |
| BAZ75991.1 | 3.74e-107 | 44 | 917 | 1148 | 2015 |
| BAY30132.1 | 1.55e-106 | 44 | 917 | 1150 | 2017 |
| BAY90071.1 | 2.37e-102 | 44 | 917 | 1147 | 2014 |
| AFY93865.1 | 8.54e-86 | 96 | 1050 | 952 | 1905 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 5BP1_A | 4.05e-151 | 105 | 973 | 10 | 865 | Condensingdi-domain (KS-AT) of a mycocerosic acid synthase-like (MAS-like) PKS [Mycolicibacterium smegmatis MC2 155] |
| 7S6B_A | 7.64e-146 | 100 | 1021 | 55 | 943 | ChainA, Polyketide synthase [Streptomyces lasalocidi],7S6B_B Chain B, Polyketide synthase [Streptomyces lasalocidi] |
| 2HG4_A | 5.10e-145 | 105 | 999 | 41 | 898 | Structureof the ketosynthase-acyltransferase didomain of module 5 from DEBS. [Saccharopolyspora erythraea],2HG4_B Structure of the ketosynthase-acyltransferase didomain of module 5 from DEBS. [Saccharopolyspora erythraea],2HG4_C Structure of the ketosynthase-acyltransferase didomain of module 5 from DEBS. [Saccharopolyspora erythraea],2HG4_D Structure of the ketosynthase-acyltransferase didomain of module 5 from DEBS. [Saccharopolyspora erythraea],2HG4_E Structure of the ketosynthase-acyltransferase didomain of module 5 from DEBS. [Saccharopolyspora erythraea],2HG4_F Structure of the ketosynthase-acyltransferase didomain of module 5 from DEBS. [Saccharopolyspora erythraea] |
| 2QO3_A | 1.23e-142 | 105 | 1006 | 8 | 889 | CrystalStructure of [KS3][AT3] didomain from module 3 of 6-deoxyerthronolide B synthase [Saccharopolyspora erythraea],2QO3_B Crystal Structure of [KS3][AT3] didomain from module 3 of 6-deoxyerthronolide B synthase [Saccharopolyspora erythraea] |
| 6C9U_A | 1.22e-141 | 98 | 1006 | 27 | 915 | Crystalstructure of [KS3][AT3] didomain from module 3 of 6-deoxyerthronolide B synthase in complex with antibody fragment (Fab) [Saccharopolyspora erythraea] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| I6X8D2 | 0.0 | 1 | 1612 | 18 | 1717 | Polyketide synthase Pks13 OS=Mycobacterium tuberculosis (strain ATCC 25618 / H37Rv) OX=83332 GN=pks13 PE=1 SV=1 |
| P9WQE7 | 3.24e-184 | 5 | 928 | 10 | 912 | Phenolphthiocerol/phthiocerol polyketide synthase subunit A OS=Mycobacterium tuberculosis (strain ATCC 25618 / H37Rv) OX=83332 GN=ppsA PE=1 SV=1 |
| P9WQE6 | 3.24e-184 | 5 | 928 | 10 | 912 | Phenolphthiocerol/phthiocerol polyketide synthase subunit A OS=Mycobacterium tuberculosis (strain CDC 1551 / Oshkosh) OX=83331 GN=ppsA PE=3 SV=1 |
| Q7TXM0 | 2.16e-182 | 5 | 928 | 10 | 912 | Phenolphthiocerol/phthiocerol polyketide synthase subunit A OS=Mycobacterium bovis (strain ATCC BAA-935 / AF2122/97) OX=233413 GN=ppsA PE=1 SV=1 |
| P9WQE4 | 1.19e-150 | 98 | 1022 | 29 | 931 | Phenolphthiocerol/phthiocerol polyketide synthase subunit B OS=Mycobacterium tuberculosis (strain CDC 1551 / Oshkosh) OX=83331 GN=ppsB PE=3 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as OTHER
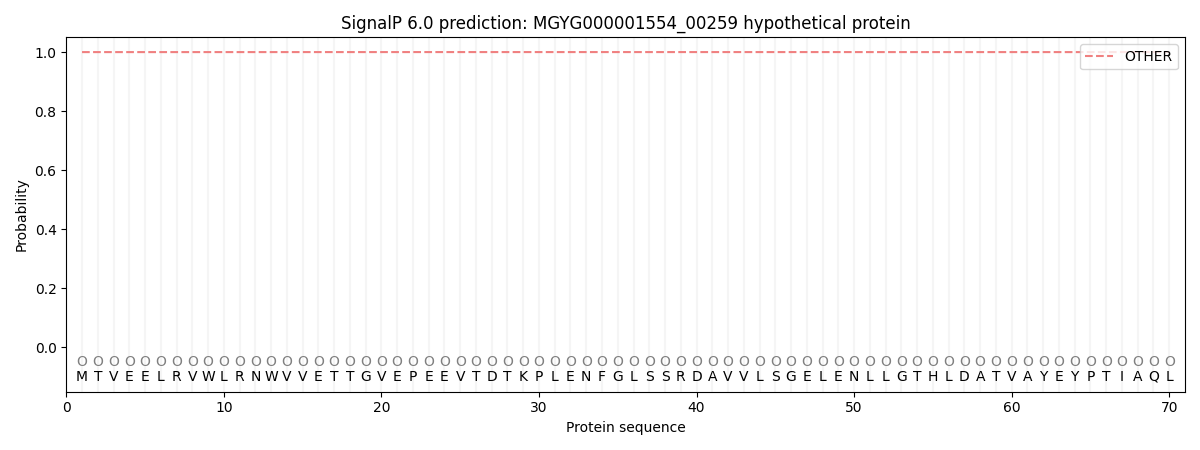
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 1.000052 | 0.000000 | 0.000000 | 0.000000 | 0.000000 | 0.000000 |
