You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000001345_00156
You are here: Home > Sequence: MGYG000001345_00156
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Bacteroides xylanisolvens | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Bacteroidota; Bacteroidia; Bacteroidales; Bacteroidaceae; Bacteroides; Bacteroides xylanisolvens | |||||||||||
| CAZyme ID | MGYG000001345_00156 | |||||||||||
| CAZy Family | GH67 | |||||||||||
| CAZyme Description | Extracellular xylan exo-alpha-(1->2)-glucuronosidase | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 60678; End: 62819 Strand: - | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH67 | 38 | 681 | 8.4e-280 | 0.9566517189835575 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| COG3661 | AguA2 | 0.0 | 19 | 689 | 3 | 682 | Alpha-glucuronidase [Carbohydrate transport and metabolism]. |
| pfam07488 | Glyco_hydro_67M | 0.0 | 113 | 435 | 1 | 324 | Glycosyl hydrolase family 67 middle domain. Alpha-glucuronidases, components of an ensemble of enzymes central to the recycling of photosynthetic biomass, remove the alpha-1,2 linked 4-O-methyl glucuronic acid from xylans. This family represents the central catalytic domain of alpha-glucuronidase. |
| pfam07477 | Glyco_hydro_67C | 3.47e-126 | 437 | 685 | 1 | 223 | Glycosyl hydrolase family 67 C-terminus. Alpha-glucuronidases, components of an ensemble of enzymes central to the recycling of photosynthetic biomass, remove the alpha-1,2 linked 4-O-methyl glucuronic acid from xylans. This family represents the C terminal region of alpha-glucuronidase which is mainly alpha-helical. It wraps around the catalytic domain (pfam07488), making additional interactions both with the N-terminal domain (pfam03648) of its parent monomer and also forming the majority of the dimer-surface with the equivalent C-terminal domain of the other monomer of the dimer. |
| pfam03648 | Glyco_hydro_67N | 5.35e-09 | 26 | 110 | 1 | 120 | Glycosyl hydrolase family 67 N-terminus. Alpha-glucuronidases, components of an ensemble of enzymes central to the recycling of photosynthetic biomass, remove the alpha-1,2 linked 4-O-methyl glucuronic acid from xylans. This family represents the N-terminal region of alpha-glucuronidase. The N-terminal domain forms a two-layer sandwich, each layer being formed by a beta sheet of five strands. A further two helices form part of the interface with the central, catalytic, module (pfam07488). |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| QUR44061.1 | 0.0 | 1 | 713 | 1 | 713 |
| CBK69516.1 | 0.0 | 1 | 713 | 1 | 713 |
| QRN00781.1 | 0.0 | 1 | 713 | 1 | 713 |
| QUT30260.1 | 0.0 | 1 | 713 | 1 | 713 |
| QUT24930.1 | 0.0 | 1 | 713 | 1 | 713 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 1GQI_A | 5.21e-246 | 21 | 692 | 1 | 683 | Structureof Pseudomonas cellulosa alpha-D-glucuronidase [Cellvibrio japonicus],1GQI_B Structure of Pseudomonas cellulosa alpha-D-glucuronidase [Cellvibrio japonicus],1GQJ_A Structure of Pseudomonas cellulosa alpha-D-glucuronidase complexed with xylobiose [Cellvibrio japonicus],1GQJ_B Structure of Pseudomonas cellulosa alpha-D-glucuronidase complexed with xylobiose [Cellvibrio japonicus],1GQK_A Structure of Pseudomonas cellulosa alpha-D-glucuronidase complexed with glucuronic acid [Cellvibrio japonicus],1GQK_B Structure of Pseudomonas cellulosa alpha-D-glucuronidase complexed with glucuronic acid [Cellvibrio japonicus],1GQL_A Structure of Pseudomonas cellulosa alpha-D-glucuronidase complexed with glucuronic acid and xylotriose [Cellvibrio japonicus],1GQL_B Structure of Pseudomonas cellulosa alpha-D-glucuronidase complexed with glucuronic acid and xylotriose [Cellvibrio japonicus] |
| 1H41_A | 4.21e-245 | 21 | 692 | 1 | 683 | Pseudomonascellulosa E292A alpha-D-glucuronidase mutant complexed with aldotriuronic acid [Cellvibrio japonicus],1H41_B Pseudomonas cellulosa E292A alpha-D-glucuronidase mutant complexed with aldotriuronic acid [Cellvibrio japonicus] |
| 1K9D_A | 6.09e-184 | 27 | 689 | 9 | 678 | The1.7 A crystal structure of alpha-D-glucuronidase, a family-67 glycoside hydrolase from Bacillus stearothermophilus T-1 [Geobacillus stearothermophilus],1L8N_A The 1.5A crystal structure of alpha-D-glucuronidase from Bacillus stearothermophilus T-1, complexed with 4-O-methyl-glucuronic acid and xylotriose [Geobacillus stearothermophilus],1MQQ_A THE CRYSTAL STRUCTURE OF ALPHA-D-GLUCURONIDASE FROM BACILLUS STEAROTHERMOPHILUS T-1 COMPLEXED WITH GLUCURONIC ACID [Geobacillus stearothermophilus] |
| 1MQP_A | 1.22e-183 | 67 | 689 | 76 | 678 | TheCrystal Structure Of Alpha-D-Glucuronidase From Bacillus Stearothermophilus T-6 [Geobacillus stearothermophilus] |
| 1MQR_A | 3.44e-183 | 67 | 689 | 76 | 678 | ChainA, ALPHA-D-GLUCURONIDASE [Geobacillus stearothermophilus] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| B3PC73 | 6.49e-245 | 21 | 692 | 25 | 707 | Extracellular xylan exo-alpha-(1->2)-glucuronosidase OS=Cellvibrio japonicus (strain Ueda107) OX=498211 GN=gla67A PE=1 SV=1 |
| Q09LY5 | 6.66e-183 | 67 | 689 | 76 | 678 | Xylan alpha-(1->2)-glucuronosidase OS=Geobacillus stearothermophilus OX=1422 GN=aguA PE=1 SV=1 |
| P96105 | 1.60e-182 | 62 | 689 | 75 | 673 | Xylan alpha-(1->2)-glucuronosidase OS=Thermotoga maritima (strain ATCC 43589 / DSM 3109 / JCM 10099 / NBRC 100826 / MSB8) OX=243274 GN=aguA PE=1 SV=2 |
| Q5AQZ4 | 5.30e-149 | 20 | 686 | 17 | 699 | Alpha-glucuronidase A OS=Emericella nidulans (strain FGSC A4 / ATCC 38163 / CBS 112.46 / NRRL 194 / M139) OX=227321 GN=aguA PE=1 SV=1 |
| B0Y2K1 | 5.88e-142 | 95 | 700 | 126 | 708 | Probable alpha-glucuronidase A OS=Neosartorya fumigata (strain CEA10 / CBS 144.89 / FGSC A1163) OX=451804 GN=aguA PE=3 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as SP
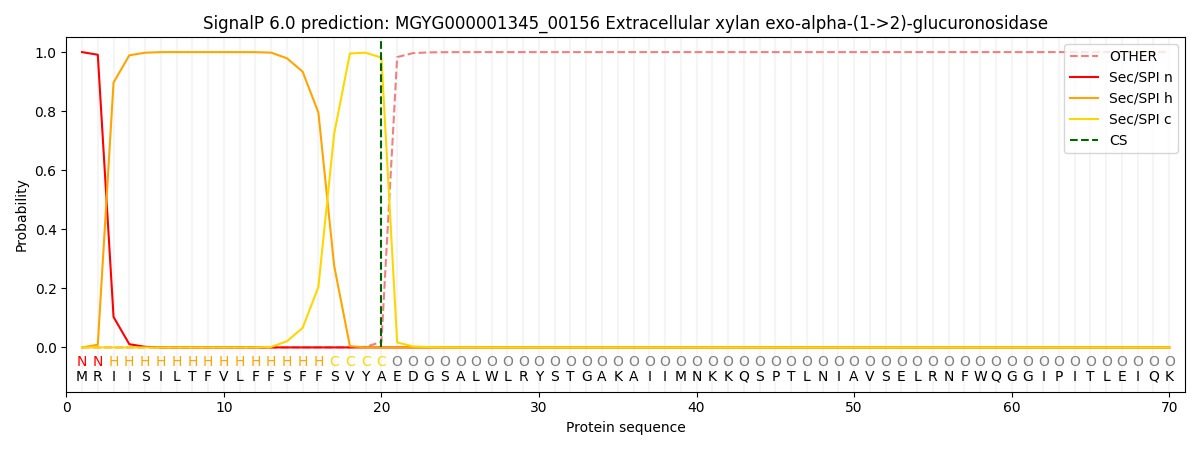
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.000448 | 0.998661 | 0.000259 | 0.000204 | 0.000204 | 0.000198 |
