You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000000501_01044
You are here: Home > Sequence: MGYG000000501_01044
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | CAG-127 sp900553925 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Firmicutes_A; Clostridia; Lachnospirales; Lachnospiraceae; CAG-127; CAG-127 sp900553925 | |||||||||||
| CAZyme ID | MGYG000000501_01044 | |||||||||||
| CAZy Family | GH73 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 95501; End: 98431 Strand: - | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH73 | 512 | 638 | 3.7e-18 | 0.9296875 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| cd02696 | MurNAc-LAA | 8.71e-54 | 244 | 435 | 1 | 172 | N-acetylmuramoyl-L-alanine amidase or MurNAc-LAA (also known as peptidoglycan aminohydrolase, NAMLA amidase, NAMLAA, Amidase 3, and peptidoglycan amidase; EC 3.5.1.28) is an autolysin that hydrolyzes the amide bond between N-acetylmuramoyl and L-amino acids in certain cell wall glycopeptides. These proteins are Zn-dependent peptidases with highly conserved residues involved in cation co-ordination. MurNAc-LAA in this family is one of several peptidoglycan hydrolases (PGHs) found in bacterial and bacteriophage or prophage genomes that are involved in the degradation of the peptidoglycan. In Escherichia coli, there are five MurNAc-LAAs present: AmiA, AmiB, AmiC and AmiD that are periplasmic, and AmpD that is cytoplasmic. Three of these (AmiA, AmiB and AmiC) belong to this family, the other two (AmiD and AmpD) do not. E. coli AmiA, AmiB and AmiC play an important role in cleaving the septum to release daughter cells after cell division. In general, bacterial MurNAc-LAAs are members of the bacterial autolytic system and carry a signal peptide in their N-termini that allows their transport across the cytoplasmic membrane. However, the bacteriophage MurNAc-LAAs are endolysins since these phage-encoded enzymes break down bacterial peptidoglycan at the terminal stage of the phage reproduction cycle. As opposed to autolysins, almost all endolysins have no signal peptides and their translocation through the cytoplasmic membrane is thought to proceed with the help of phage-encoded holin proteins. The amidase catalytic module is fused to another functional module (cell wall binding module or CWBM) either at the N- or C-terminus, which is responsible for high affinity binding of the protein to the cell wall. |
| pfam01520 | Amidase_3 | 4.30e-45 | 245 | 434 | 1 | 172 | N-acetylmuramoyl-L-alanine amidase. This enzyme domain cleaves the amide bond between N-acetylmuramoyl and L-amino acids in bacterial cell walls. |
| COG0860 | AmiC | 1.18e-39 | 244 | 440 | 44 | 230 | N-acetylmuramoyl-L-alanine amidase [Cell wall/membrane/envelope biogenesis]. |
| TIGR02883 | spore_cwlD | 7.26e-30 | 244 | 437 | 2 | 189 | N-acetylmuramoyl-L-alanine amidase CwlD. Members of this protein family are the CwlD family of N-acetylmuramoyl-L-alanine amidase. This family has been called the germination-specific N-acetylmuramoyl-L-alanine amidase. CwlD is required, along with the putative deactylase PdaA, to make muramic delta-lactam, a novel peptidoglycan constituent found only in spores. CwlD mutants show a germination defect. [Cell envelope, Biosynthesis and degradation of murein sacculus and peptidoglycan, Cellular processes, Sporulation and germination] |
| COG5492 | YjdB | 3.32e-19 | 646 | 907 | 14 | 306 | Uncharacterized conserved protein YjdB, contains Ig-like domain [General function prediction only]. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| QNM00238.1 | 0.0 | 1 | 976 | 1 | 950 |
| VCV20908.1 | 4.03e-70 | 70 | 442 | 170 | 545 |
| CBL07763.1 | 7.16e-70 | 70 | 442 | 164 | 539 |
| CBL13779.1 | 1.28e-69 | 70 | 442 | 165 | 540 |
| QRO35993.1 | 1.66e-69 | 58 | 443 | 93 | 479 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 5J72_A | 3.34e-20 | 245 | 427 | 454 | 626 | ChainA, Putative N-acetylmuramoyl-L-alanine amidase,autolysin cwp6 [Clostridioides difficile 630],5J72_B Chain B, Putative N-acetylmuramoyl-L-alanine amidase,autolysin cwp6 [Clostridioides difficile 630] |
| 5EMI_A | 2.68e-15 | 243 | 437 | 5 | 177 | ChainA, Cell wall hydrolase/autolysin [Nostoc punctiforme PCC 73102] |
| 4BIN_A | 8.32e-14 | 243 | 345 | 174 | 272 | Crystalstructure of the E. coli N-acetylmuramoyl-L-alanine amidase AmiC [Escherichia coli K-12] |
| 3NE8_A | 1.25e-13 | 240 | 331 | 2 | 97 | Thecrystal structure of a domain from N-acetylmuramoyl-l-alanine amidase of Bartonella henselae str. Houston-1 [Bartonella henselae] |
| 4RN7_A | 3.17e-13 | 245 | 436 | 6 | 176 | ChainA, N-acetylmuramoyl-L-alanine amidase [Clostridioides difficile 630] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| P37134 | 2.85e-23 | 245 | 483 | 4 | 216 | N-acetylmuramoyl-L-alanine amidase CwlM OS=Bacillus licheniformis OX=1402 GN=cwlM PE=3 SV=1 |
| O48471 | 8.77e-22 | 244 | 442 | 4 | 185 | Endolysin OS=Bacillus phage SPP1 OX=10724 GN=25 PE=4 SV=1 |
| Q06320 | 2.11e-21 | 245 | 447 | 4 | 184 | Sporulation-specific N-acetylmuramoyl-L-alanine amidase OS=Bacillus subtilis (strain 168) OX=224308 GN=cwlC PE=1 SV=1 |
| O51481 | 4.79e-20 | 508 | 609 | 68 | 168 | Uncharacterized protein BB_0531 OS=Borreliella burgdorferi (strain ATCC 35210 / DSM 4680 / CIP 102532 / B31) OX=224326 GN=BB_0531 PE=3 SV=1 |
| Q02114 | 3.62e-16 | 245 | 439 | 322 | 490 | N-acetylmuramoyl-L-alanine amidase LytC OS=Bacillus subtilis (strain 168) OX=224308 GN=lytC PE=1 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as SP
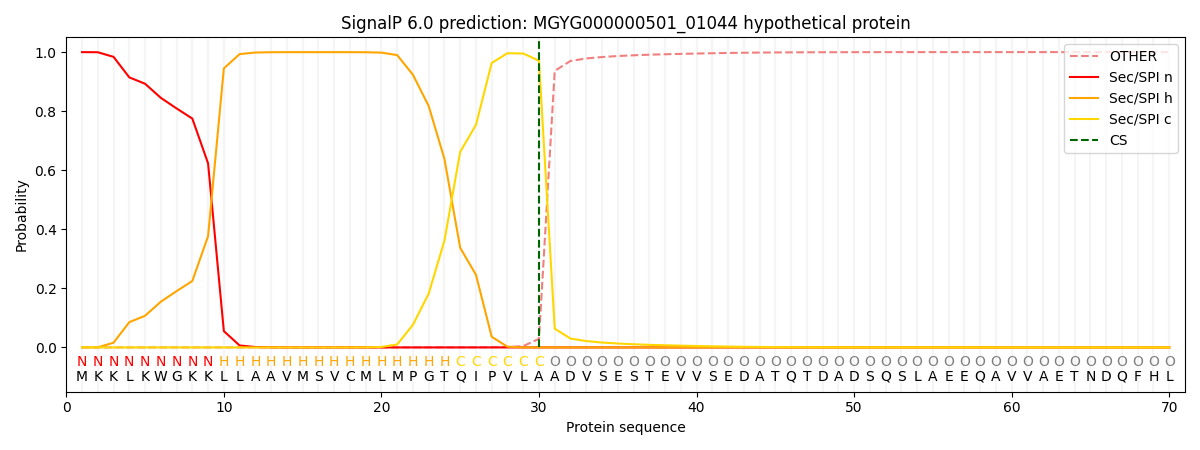
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.000431 | 0.998624 | 0.000322 | 0.000214 | 0.000190 | 0.000167 |
